Mesenchymal stem cell-based therapy: a new paradigm in regenerative medicine
- PMID: 19602034
- PMCID: PMC4515054
- DOI: 10.1111/j.1582-4934.2009.00857.x
Mesenchymal stem cell-based therapy: a new paradigm in regenerative medicine
Abstract
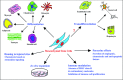
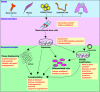
Mesenchymal stem cells (MSCs), adherent fibroblastoid cells, present in bone marrow and many other tissues can be easily isolated and expanded in vitro. They are capable of differentiating into different cell types such as osteoblasts, chondrocytes, adipocytes, cardiomyocytes, hepatocytes, endothelial cells and neuronal cells. Such immense plasticity coupled with their ability to modulate the activity of immune cells makes them attractive for stem cell-based therapy aimed at treating previously incurable disorders. Preclinical studies have reported successful use of MSCs for delivering therapeutic proteins and repairing defects in a variety of disease models. These studies highlighted the in vivo potential of MSCs and their ability to home to injury sites and modify the microenvironment by secreting paracrine factors to augment tissue repair. Their therapeutic applicability has been widened by genetic modification to enhance differentiation and tissue targeting, and use in tissue engineering. Clinical trials for diseases such as osteogenesis imperfecta, graft-versus-host disease and myocardial infarction have shown some promise, demonstrating the safe use of both allogeneic and autologous cells. However, lack of knowledge of MSC behaviour and responses in vitro and in vivo force the need for basic and animal studies before heading to the clinic. Contrasting reports on immunomodulatory functions and tumorigenicity along with issues such as mode of cell delivery, lack of specific marker, low survival and engraftment require urgent attention to harness the potential of MSC-based therapy in the near future.
Figures
References
-
- Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–7. - PubMed
-
- Bruder SP, Jaiswal N, Haynesworth SE. Growth kinetics, self-renewal and the osteogenic potential of purified human mesenchymal stem cells during extensive subcultivation and following cryipreservation. J Cell Biochem. 1997;64:278–94. - PubMed
-
- Horwitz ED, Prockop DJ, Fitzpatrick LA, et al. Transplantability and therapeutic effects of bone marrow-derived mesenchymal cells in children with osteogenesis imperfecta. Nat Med. 1999;5:309–13. - PubMed
-
- Chen SL, Fang WW, Ye F, et al. Effect on left ventricular function of intracoronary transplantation of autologous bone marrow mesenchymal stem cell in patients with acute myocardial infarction. Am J Cardiol. 2004;94:92–5. - PubMed
-
- Bang OY, Lee JS, Lee PH, et al. Autologous mesenchymal stem cell transplantation in stroke patients. Ann Neurol. 2005;57:874–82. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

