Exploring symmetry-based logic for a synthesis of palau'amine
- PMID: 19603820
- PMCID: PMC2888864
- DOI: 10.1021/jo900755r
Exploring symmetry-based logic for a synthesis of palau'amine
Abstract
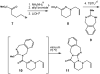
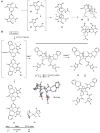
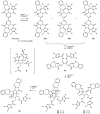
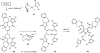
A synthetic approach to palau'amine is described that exploits veiled symmetry in the structure. Bis-alkylidenes i have been prepared and found susceptible to halogenative desymmetrization using t-BuOCl. This oxidation forms the imbedded spirocyclopentane motif observed in the natural product. A host of atypical reactions and processes developed during these studies are discussed, as are plans for completing total syntheses of this compound class.
Figures





References
-
- Kinnel RB, Gehrken H-P, Scheuer PJ. J. Am. Chem. Soc. 1993;115:3376–3377.
- Kinnel RB, Gehrken H-P, Swali R, Skoropowski G, Scheuer PJ. J. Org. Chem. 1998;63:3281–3286.
-
- Wright AE, Chiles SA, Cross SS. J. Nat. Prod. 1991;54:1684–1686. - PubMed
-
- Garcia EE, Benjamin LE, Fryer RI. J. Chem. Soc., Chem. Comm. 1973:78–79.
- Gautschi JT, Whitman S, Holman TR, Crews P. J. Nat. Prd. 2004;67:1256–1261. - PubMed
-
- Ravert N, Al-Mourabit A. J. Am. Chem. Soc. 2004;126:10252–10253. - PubMed
- Vergne C, Boury-Esnault N, Perez T, Martin M-T, Adeline M-T, Dau ETH, Al-Mourabit A. Org. Lett. 2006;8:2421–2424. - PubMed
- Vergne C, Appenzeller J, Ratinaud C, Martin M-T, Debitus C, Zaparucha A, Al-Mourabit A. Org. Lett. 2008;10:493–496. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

