Molecular mechanisms of 4-hydroxy-2-nonenal and acrolein toxicity: nucleophilic targets and adduct formation
- PMID: 19610654
- PMCID: PMC4452948
- DOI: 10.1021/tx900147g
Molecular mechanisms of 4-hydroxy-2-nonenal and acrolein toxicity: nucleophilic targets and adduct formation
Abstract
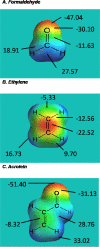
Acrolein and 4-hydroxy-2-nonenal (HNE) are byproducts of lipid peroxidation and are thought to play central roles in various traumatic injuries and disease states that involve cellular oxidative stress, for example, spinal cord trauma, diabetes, and Alzheimer's disease. In this review, we will discuss the chemical attributes of acrolein and HNE that determine their toxicities. Specifically, these aldehydes are classified as type 2 alkenes and are characterized by an alpha,beta-unsaturated carbonyl structure. This structure is a conjugated system that contains mobile pi-electrons. The carbonyl oxygen atom is electronegative and can promote the withdrawal of mobile electron density from the beta-carbon atom causing regional electron deficiency. On the basis of this type of electron polarizability, both acrolein and HNE are considered to be soft electrophiles that preferentially form 1,4-Michael type adducts with soft nucleophiles. Proteomic, quantum mechanical, and kinetic data will be presented, indicating that cysteine sulfhydryl groups are the primary soft nucleophilic targets of acrolein and HNE. This is in contrast to nitrogen groups on harder biological nucleophiles such as lysine or histidine residues. The toxicological outcome of adduct formation is not only dependent upon residue selectivity but also the importance of the targeted amino acid in protein function or structure. In attempting to discern the toxicological significance of a given adduct, we will consider the normal roles of cysteine, lysine, and histidine residues in proteins and the relative merits of corresponding adducts in the manifestations of diseases or toxic states. Understanding the molecular actions of acrolein and HNE could provide insight into many pathogenic conditions that involve initial cellular oxidative stress and could, thereby, offer new efficacious avenues of pharmacological defense.
Figures
References
-
- Esterbauer H, Schaur RJ, Zollner J. Chemistry and biochemistry of 4-hydroxynonenal, malonaldehyde and related aldehydes. Free Rad. Biol. Med. 1991;11:81–128. - PubMed
-
- Halliwell B. Oxidative stress and neurodegeneration: where are we now? J. Neurochem. 2006;97:1634–1658. - PubMed
-
- Lefer DJ, Granger N. Oxidative stress and cardiac disease. Am. J. Med. 2000;109:316–323. - PubMed
-
- Lieber CS. Alcoholic liver injury: pathogenesis and therapy in 2001. Pathol. Biol. 2001;49:738–752. - PubMed
-
- Povlishock JT, Kontos HA. The role of oxygen radicals in the pathobiology of traumatic brain injury. Hum Cell. 1992;5:345–353. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

