Mu opioid receptor modulation of somatodendritic dopamine overflow: GABAergic and glutamatergic mechanisms
- PMID: 19614973
- PMCID: PMC2761213
- DOI: 10.1111/j.1460-9568.2009.06827.x
Mu opioid receptor modulation of somatodendritic dopamine overflow: GABAergic and glutamatergic mechanisms
Abstract
Mu opioid receptor (MOR) regulation of somatodendritic dopamine neurotransmission in the ventral tegmental area (VTA) was investigated using conventional microdialysis in freely moving rats and mice. Reverse dialysis of the MOR agonist DAMGO (50 and 100 microm) into the VTA of rats produced a concentration-dependent increase in dialysate dopamine concentrations. Basal dopamine overflow in the VTA was unaltered in mice lacking the MOR gene. However, basal gamma-aminobutyric acid (GABA) overflow in these animals was significantly increased, whereas glutamate overflow was decreased. Intra-VTA perfusion of DAMGO into wild-type (WT) mice increased dopamine overflow. GABA concentrations were decreased, whereas glutamate concentrations in the VTA were unaltered. Consistent with the loss of MOR, no effect of DAMGO was observed in MOR knockout (KO) mice. These data provide the first direct demonstration of tonically active MOR systems in the VTA that regulate basal glutamatergic and GABAergic neurotransmission in this region. We hypothesize that increased GABAergic neurotransmission following constitutive deletion of MOR is due to the elimination of a tonic inhibitory influence of MOR on GABAergic neurons in the VTA, whereas decreased glutamatergic neurotransmission in MOR KO mice is a consequence of intensified GABA tone on glutamatergic neurons and/or terminals. As a consequence, somatodendritic dopamine release is unaltered. Furthermore, MOR KO mice do not exhibit the positive correlation between basal dopamine levels and the glutamate/GABA ratio observed in WT mice. Together, our findings indicate a critical role of VTA MOR in maintaining an intricate balance between excitatory and inhibitory inputs to dopaminergic neurons.
Figures
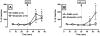
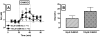




References
-
- Adell A, Artigas F. The somatodendritic release of dopamine in the ventral tegmental area and its regulation by afferent transmitter systems. Neurosci Biobehav Rev. 2004;28(4):415–431. - PubMed
-
- Bals-Kubik R, Ableitner A, Herz A, Shippenberg TS. Neuroanatomical sites mediating the motivational effects of opioids as mapped by the conditioned place preference paradigm in rats. J Pharmacol Exp Ther. 1993;264:489–495. - PubMed
-
- Bergevin A, Girardot D, Bourque MJ, Trudeau LE. Presynaptic mu-opioid receptors regulate a late step of the secretory process in rat ventral tegmental area GABAergic neurons. Neuropharmacology. 2002;42:1065–1078. - PubMed
-
- Bozarth MA, Wise RA. Involvement of the ventral tegmental dopamine system in opioid and psychomotor stimulant reinforcement. NIDA Res Monogr. 1986;67:190–196. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

