Role of the bed nucleus of the stria terminalis in the control of ventral tegmental area dopamine neurons
- PMID: 19616054
- PMCID: PMC3635540
- DOI: 10.1016/j.pnpbp.2009.07.010
Role of the bed nucleus of the stria terminalis in the control of ventral tegmental area dopamine neurons
Abstract
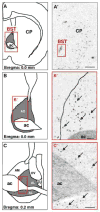
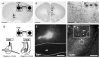
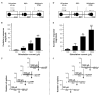
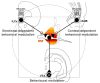
Projections from neurons of the bed nucleus of the stria terminalis (BST) to the ventral tegmental area (VTA) are crucial to behaviors related to reward and motivation. Over the past few years, we have undertaken a series of studies to understand: 1) how excitatory inputs regulate in vivo excitable properties of BST neurons, and 2) how BST inputs in turn modulate neuronal activity of dopamine neurons in VTA. Using in vivo extracellular recording techniques in anesthetized rats and tract-tracing approaches, we have demonstrated that inputs from the infralimbic cortex and the ventral subiculum exert a strong excitatory influence on BST neurons projecting to the VTA. Thus, the BST is uniquely positioned to receive emotional and learning-associated informations and to integrate these into the reward/motivation circuitry. We will discuss how changes in the activity of BST neurons projecting to the VTA could participate in the development or exacerbation of psychiatric conditions such as drug addiction.
Figures



Similar articles
-
Maternally responsive neurons in the bed nucleus of the stria terminalis and medial preoptic area: Putative circuits for regulating anxiety and reward.Front Neuroendocrinol. 2015 Jul;38:65-72. doi: 10.1016/j.yfrne.2015.04.001. Epub 2015 Apr 21. Front Neuroendocrinol. 2015. PMID: 25910426 Free PMC article. Review.
-
GABAergic neurons in the ventral tegmental area receive dual GABA/enkephalin-mediated inhibitory inputs from the bed nucleus of the stria terminalis.Eur J Neurosci. 2014 Jun;39(11):1796-809. doi: 10.1111/ejn.12503. Epub 2014 Mar 2. Eur J Neurosci. 2014. PMID: 24580812
-
Three types of neurochemical projection from the bed nucleus of the stria terminalis to the ventral tegmental area in adult mice.J Neurosci. 2012 Dec 12;32(50):18035-46. doi: 10.1523/JNEUROSCI.4057-12.2012. J Neurosci. 2012. PMID: 23238719 Free PMC article.
-
Activation of ventral tegmental area cells by the bed nucleus of the stria terminalis: a novel excitatory amino acid input to midbrain dopamine neurons.J Neurosci. 2002 Jun 15;22(12):5173-87. doi: 10.1523/JNEUROSCI.22-12-05173.2002. J Neurosci. 2002. PMID: 12077212 Free PMC article.
-
Ethanol action on dopaminergic neurons in the ventral tegmental area: interaction with intrinsic ion channels and neurotransmitter inputs.Int Rev Neurobiol. 2010;91:235-88. doi: 10.1016/S0074-7742(10)91008-8. Int Rev Neurobiol. 2010. PMID: 20813245 Free PMC article. Review.
Cited by
-
Optogenetic strategies to investigate neural circuitry engaged by stress.Behav Brain Res. 2013 Oct 15;255:19-25. doi: 10.1016/j.bbr.2013.05.007. Epub 2013 May 16. Behav Brain Res. 2013. PMID: 23684554 Free PMC article. Review.
-
Ethanol actions on the ventral tegmental area: novel potential targets on reward pathway neurons.Psychopharmacology (Berl). 2018 Jun;235(6):1711-1726. doi: 10.1007/s00213-018-4875-y. Epub 2018 Mar 16. Psychopharmacology (Berl). 2018. PMID: 29549390 Free PMC article. Review.
-
Maternally responsive neurons in the bed nucleus of the stria terminalis and medial preoptic area: Putative circuits for regulating anxiety and reward.Front Neuroendocrinol. 2015 Jul;38:65-72. doi: 10.1016/j.yfrne.2015.04.001. Epub 2015 Apr 21. Front Neuroendocrinol. 2015. PMID: 25910426 Free PMC article. Review.
-
The Proinflammatory Cytokine Interleukin 18 Regulates Feeding by Acting on the Bed Nucleus of the Stria Terminalis.J Neurosci. 2016 May 4;36(18):5170-80. doi: 10.1523/JNEUROSCI.3919-15.2016. J Neurosci. 2016. PMID: 27147667 Free PMC article.
-
Neurotensin immunolabeling relates to sexually-motivated song and other social behaviors in male European starlings (Sturnus vulgaris).Behav Brain Res. 2015 Apr 1;282:133-43. doi: 10.1016/j.bbr.2015.01.007. Epub 2015 Jan 13. Behav Brain Res. 2015. PMID: 25595421 Free PMC article.
References
-
- Aston-Jones G, Harris GC. Brain substrates for increased drug seeking during protracted withdrawal. Neuropharmacology. 2004;47:167–179. - PubMed
-
- Aston-Jones G, Delfs JM, Druhan J, Zhu Y. The bed nucleus of the stria terminalis. A target site for noradrenergic actions in opiate withdrawal. Ann N Y Acad Sci. 1999;877:486–498. - PubMed
-
- Blaha CD, Yang CR, Floresco SB, Barr AM, Phillips AG. Stimulation of the ventral subiculum of the hippocampus evokes glutamate receptor-mediated changes in dopamine efflux in the rat nucleus accumbens. Eur J Neurosci. 1997;9:902–911. - PubMed
-
- Bowers WJ, Attiast E, Amit Z. Stress enhances the response to reward reduction but not food-motivated responding. Physiol Behav. 1999;67:777–782. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

