Inhibition of CXCR2 signaling promotes recovery in models of multiple sclerosis
- PMID: 19616545
- PMCID: PMC2761527
- DOI: 10.1016/j.expneurol.2009.07.010
Inhibition of CXCR2 signaling promotes recovery in models of multiple sclerosis
Abstract
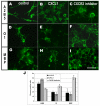
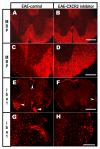
Multiple sclerosis (MS) is a neurodegenerative disease characterized by demyelination/remyelination episodes that ultimately fail. Chemokines and their receptors have been implicated in both myelination and remyelination failure. Chemokines regulate migration, proliferation and differentiation of immune and neural cells during development and pathology. Previous studies have demonstrated that the absence of the chemokine receptor CXCR2 results in both disruption of early oligodendrocyte development and long-term structural alterations in myelination. Histological studies suggest that CXCL1, the primary ligand for CXCR2, is upregulated around the peripheral areas of demyelination suggesting that this receptor/ligand combination modulates responses to injury. Here we show that in focal LPC-induced demyelinating lesions, localized inhibition of CXCR2 signaling reduced lesion size and enhanced remyelination while systemic treatments were relatively less effective. Treatment of spinal cord cultures with CXCR2 antagonists reduced CXCL1 induced A2B5+ cell proliferation and increased differentiation of myelin producing cells. More critically, treatment of myelin oligodendrocyte glycoprotein peptide 35-55-induced EAE mice, an animal model of multiple sclerosis, with small molecule antagonists against CXCR2 results in increased functionality, decreased lesion load, and enhanced remyelination. Our findings demonstrate the importance of antagonizing CXCR2 in enhancing myelin repair by reducing lesion load and functionality in models of multiple sclerosis and thus providing a therapeutic target for demyelinating diseases.
Figures






References
-
- Antel J. Multiple sclerosis--emerging concepts of disease pathogenesis. J Neuroimmunol. 1999;98:45–48. - PubMed
-
- Bajetto A, Bonavia R, Barbero S, Florio T, Schettini G. Chemokines and their receptors in the central nervous system. Front Neuroendocrinol. 2001;22:147–184. - PubMed
-
- Bambakidis NC, Wang RZ, Franic L, Miller RH. Sonic hedgehog-induced neural precursor proliferation after adult rodent spinal cord injury. J Neurosurg. 2003;99:70–75. - PubMed
-
- Barnes PJ. Cytokine modulators as novel therapies for airway disease. Eur Respir J Suppl. 2001;34:67s–77s. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

