MCP-1 antibody treatment enhances damage and impedes repair of the alveolar epithelium in influenza pneumonitis
- PMID: 19617401
- PMCID: PMC2891499
- DOI: 10.1165/rcmb.2008-0423OC
MCP-1 antibody treatment enhances damage and impedes repair of the alveolar epithelium in influenza pneumonitis
Abstract
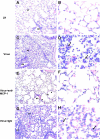
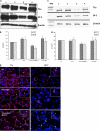
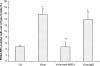
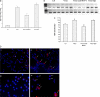
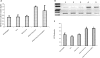
Recent studies have demonstrated an essential role of alveolar macrophages during influenza virus infection. Enhanced mortalities were observed in macrophage-depleted mice and pigs after influenza virus infection, but the basis for the enhanced pathogenesis is unclear. This study revealed that blocking macrophage recruitment into the lungs in a mouse model of influenza pneumonitis resulted in enhanced alveolar epithelial damage and apoptosis, as evaluated by histopathology, immunohistochemistry, Western blot, RT-PCR, and TUNEL assays. Abrogation of macrophage recruitment was achieved by treatment with monoclonal antibody against monocyte chemoattractant protein-1 (MCP-1) after sub-lethal challenge with mouse-adapted human influenza A/Aichi/2/68 virus. Interestingly, elevated levels of hepatocyte growth factor (HGF), a mitogen for alveolar epithelium, were detected in bronchoalveolar lavage samples and in lung homogenates of control untreated and nonimmune immunoglobulin (Ig)G-treated mice after infection compared with anti-MCP-1-treated infected mice. The lungs of control animals also displayed strongly positive HGF staining in alveolar macrophages as well as alveolar epithelial cell hyperplasia. Co-culture of influenza virus-infected alveolar epithelial cells with freshly isolated alveolar macrophages induced HGF production and phagocytic activity of macrophages. Recombinant HGF added to mouse lung explants after influenza virus infection resulted in enhanced BrdU labeling of alveolar type II epithelial cells, indicating their proliferation, in contrast with anti-HGF treatment showing significantly reduced epithelial regeneration. Our data indicate that inhibition of macrophage recruitment augmented alveolar epithelial damage and apoptosis during influenza pneumonitis, and that HGF produced by macrophages in response to influenza participates in the resolution of alveolar epithelium.
Figures

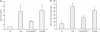


References
-
- Chow VT, Tambyah PA, Goh KT. To kill a mocking bird flu? Ann Acad Med Singapore 2008;37:451–453. - PubMed
-
- Lal SK, Chow VT. Avian influenza H5N1 virus: an emerging global pandemic. In: Lal SK, editor. Emerging viral diseases of Southeast Asia. Basel: Karger; 2007. pp. 59–77.
-
- Russell CA, Jones TC, Barr IG, Cox NJ, Garten RJ, Gregory V, Gust ID, Hampson AW, Hay AJ, Hurt AC, et al. The global circulation of seasonal influenza A (H3N2) viruses. Science 2008;320:340–346. - PubMed
-
- Novel Swine-Origin Influenza A (H1N1) Virus Investigation Team. Emergence of a novel swine-origin influenza A (H1N1) virus in humans. N Engl J Med 2009;360:2605–2615. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

