Ect2 links the PKCiota-Par6alpha complex to Rac1 activation and cellular transformation
- PMID: 19617897
- PMCID: PMC2762483
- DOI: 10.1038/onc.2009.217
Ect2 links the PKCiota-Par6alpha complex to Rac1 activation and cellular transformation
Abstract
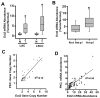
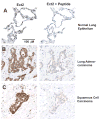
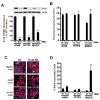
Protein kinase Ciota (PKCiota) promotes non-small cell lung cancer (NSCLC) by binding to Par6alpha and activating a Rac1-Pak-Mek1,2-Erk1,2 signaling cascade. The mechanism by which the PKCiota-Par6alpha complex regulates Rac1 is unknown. Here we show that epithelial cell transforming sequence 2 (Ect2), a guanine nucleotide exchange factor for Rho family GTPases, is coordinately amplified and overexpressed with PKCiota in NSCLC tumors. RNA interference-mediated knockdown of Ect2 inhibits Rac1 activity and blocks transformed growth, invasion and tumorigenicity of NSCLC cells. Expression of constitutively active Rac1 (RacV12) restores transformation to Ect2-deficient cells. Interestingly, the role of Ect2 in transformation is distinct from its well-established role in cytokinesis. In NSCLC cells, Ect2 is mislocalized to the cytoplasm where it binds the PKCiota-Par6alpha complex. RNA interference-mediated knockdown of either PKCiota or Par6alpha causes Ect2 to redistribute to the nucleus, indicating that the PKCiota-Par6alpha complex regulates the cytoplasmic localization of Ect2. Our data indicate that Ect2 and PKCiota are genetically and functionally linked in NSCLC, acting to coordinately drive tumor cell proliferation and invasion through formation of an oncogenic PKCiota-Par6alpha-Ect2 complex.
Figures
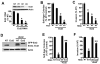


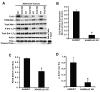
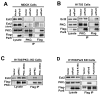
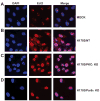
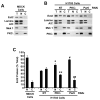
Similar articles
-
Oncogenic activity of Ect2 is regulated through protein kinase C iota-mediated phosphorylation.J Biol Chem. 2011 Mar 11;286(10):8149-8157. doi: 10.1074/jbc.M110.196113. Epub 2010 Dec 28. J Biol Chem. 2011. PMID: 21189248 Free PMC article.
-
Matrix metalloproteinase-10 is a critical effector of protein kinase Ciota-Par6alpha-mediated lung cancer.Oncogene. 2008 Aug 14;27(35):4841-53. doi: 10.1038/onc.2008.119. Epub 2008 Apr 21. Oncogene. 2008. PMID: 18427549 Free PMC article.
-
Protein kinase Cι promotes UBF1-ECT2 binding on ribosomal DNA to drive rRNA synthesis and transformed growth of non-small-cell lung cancer cells.J Biol Chem. 2020 Jun 12;295(24):8214-8226. doi: 10.1074/jbc.RA120.013175. Epub 2020 Apr 29. J Biol Chem. 2020. PMID: 32350115 Free PMC article.
-
Oncogenic Ect2 signaling regulates rRNA synthesis in NSCLC.Small GTPases. 2019 Sep;10(5):388-394. doi: 10.1080/21541248.2017.1335274. Epub 2017 Jul 5. Small GTPases. 2019. PMID: 28657426 Free PMC article. Review.
-
Targeting the oncogenic protein kinase Ciota signalling pathway for the treatment of cancer.Biochem Soc Trans. 2007 Nov;35(Pt 5):996-1000. doi: 10.1042/BST0350996. Biochem Soc Trans. 2007. PMID: 17956262 Review.
Cited by
-
Amplified genes may be overexpressed, unchanged, or downregulated in cervical cancer cell lines.PLoS One. 2012;7(3):e32667. doi: 10.1371/journal.pone.0032667. Epub 2012 Mar 7. PLoS One. 2012. PMID: 22412903 Free PMC article.
-
14-3-3ζ and aPKC-ι synergistically facilitate epithelial-mesenchymal transition of cholangiocarcinoma via GSK-3β/Snail signaling pathway.Oncotarget. 2016 Aug 23;7(34):55191-55210. doi: 10.18632/oncotarget.10483. Oncotarget. 2016. PMID: 27409422 Free PMC article.
-
Copy number gains of FGFR1 and 3q chromosome in squamous cell carcinoma of the lung.Transl Lung Cancer Res. 2013 Apr;2(2):101-11. doi: 10.3978/j.issn.2218-6751.2013.03.05. Transl Lung Cancer Res. 2013. PMID: 25806221 Free PMC article. Review.
-
Polyploid cardiomyocytes: implications for heart regeneration.Development. 2021 Jul 15;148(14):dev199401. doi: 10.1242/dev.199401. Epub 2021 Jul 26. Development. 2021. PMID: 34897388 Free PMC article.
-
Integration of Chemoinformatics and Multi-Omics Analysis Defines ECT2 as a Potential Target for Cancer Drug Therapy.Biology (Basel). 2023 Apr 18;12(4):613. doi: 10.3390/biology12040613. Biology (Basel). 2023. PMID: 37106813 Free PMC article.
References
-
- Balsara BR, Sonoda G, du Manoir S, Siegfried JM, Gabrielson E, Testa JR. Comparative genomic hybridization analysis detects frequent, often high-level, overrepresentation of DNA sequences at 3q, 5p, 7p, and 8q in human non-small cell lung carcinomas. Cancer Res. 1997;57:2116–20. - PubMed
-
- Brass N, Ukena I, Remberger K, Mack U, Sybrecht GW, Meese EU. DNA amplification on chromosome 3q26.1-q26.3 in squamous cell carcinoma of the lung detected by reverse chromosome painting. Eur J Cancer. 1996;32A:1205–8. - PubMed
-
- Hara T, Abe M, Inoue H, Yu LR, Veenstra TD, Kang YH, et al. Cytokinesis regulator ECT2 changes its conformation through phosphorylation at Thr-341 in G2/M phase. Oncogene. 2006;25:566–78. - PubMed
-
- Hirata D, Yamabuki T, Miki D, Ito T, Tsuchiya E, Fujita M, et al. Involvement of Epithelial Cell Transforming Sequence-2 Oncoantigen in Lung and Esophageal Cancer Progression. Clin Cancer Res. 2009;15:256–266. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

