KSP inhibitor ARRY-520 as a substitute for Paclitaxel in Type I ovarian cancer cells
- PMID: 19619321
- PMCID: PMC2719595
- DOI: 10.1186/1479-5876-7-63
KSP inhibitor ARRY-520 as a substitute for Paclitaxel in Type I ovarian cancer cells
Abstract
Background: We previously described a sub-population of epithelial ovarian cancer (EOC) cells with a functional TLR-4/MyD88/NF-kappaB pathway (Type I EOC cells), which confers the capacity to respond to Paclitaxel, a known TLR-4 ligand, by enhancing NF-kappaB activity and upregulating cytokine secretion - events that are known to promote tumor progression. It is therefore important to distinguish those patients that should not receive Paclitaxel; it is also important to identify alternative chemotherapy options that would benefit this sub-group of patients. The objective of this study is to determine if the KSP inhibitor, ARRY-520, can be a substitute for Paclitaxel in patients with Type I EOC.
Methods: EOC cells isolated from either ascites or tumor tissue were treated with increasing concentrations of ARRY-520 or Paclitaxel and cell viability determined. Activation of the apoptotic pathway was determined using Western blot analysis. Mitochondrial integrity was quantified using JC1 dye. Cytokine profiling was performed from supernatants using xMAP technology. NF-kappaB activity was measured using a Luciferase reporter system. In vivo activity was determined using a subcutaneous xenograft mouse model.
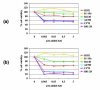
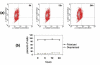
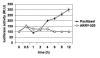
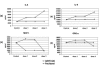
Results: ARRY-520 and Paclitaxel exhibited the same cytotoxic effect on Type I and II cells. The GI50 at 48 h for Type II EOC cells was 0.0015 microM and 0.2 microM for ARRY-520 and Paclitaxel, respectively. For Type I EOC cells, the GI50 at 48 h was > 3 microM and >20 microM for ARRY-520 and Paclitaxel, respectively. Decrease in the number of viable cells was accompanied by mitochondrial depolarization and caspase activation. Unlike Paclitaxel, ARRY-520 did not induce NF-kappaB activation, did not enhance cytokine secretion, nor induce ERK phosphorylation in Type I EOC cells.
Conclusion: Administration of Paclitaxel to patients with high percentage Type I cancer cells could have detrimental effects due to Paclitaxel-induced enhancement of NF-kappaB and ERK activities, and cytokine production (e.g. IL-6), which promote chemoresistance and tumor progression. ARRY-520 has similar anti-tumor activity in EOC cells as that of Paclitaxel. However, unlike Paclitaxel, it does not induce these pro-tumor effects in Type I cells. Therefore, the KSP inhibitor ARRY-520 may represent an alternative to Paclitaxel in this subgroup of EOC patients.
Figures



Similar articles
-
NSAID-activated gene 1 mediates pro-inflammatory signaling activation and paclitaxel chemoresistance in type I human epithelial ovarian cancer stem-like cells.Oncotarget. 2016 Nov 1;7(44):72148-72166. doi: 10.18632/oncotarget.12355. Oncotarget. 2016. PMID: 27708225 Free PMC article.
-
Differential activation of NF-κB signaling is associated with platinum and taxane resistance in MyD88 deficient epithelial ovarian cancer cells.Int J Biochem Cell Biol. 2015 Apr;61:90-102. doi: 10.1016/j.biocel.2015.02.001. Epub 2015 Feb 11. Int J Biochem Cell Biol. 2015. PMID: 25681684
-
Atractylenolide-I sensitizes human ovarian cancer cells to paclitaxel by blocking activation of TLR4/MyD88-dependent pathway.Sci Rep. 2014 Jan 23;4:3840. doi: 10.1038/srep03840. Sci Rep. 2014. PMID: 24452475 Free PMC article.
-
Expert consensus: Profiling and management of advanced or metastatic epithelial ovarian cancer.Rev Colomb Obstet Ginecol. 2024 Jun 14;75(1):4094. doi: 10.18597/rcog.4094. Rev Colomb Obstet Ginecol. 2024. PMID: 39013199 Free PMC article. English, Spanish.
-
Dose-dense paclitaxel in advanced ovarian cancer.Clin Oncol (R Coll Radiol). 2015 Jan;27(1):40-7. doi: 10.1016/j.clon.2014.10.001. Epub 2014 Nov 4. Clin Oncol (R Coll Radiol). 2015. PMID: 25455846 Review.
Cited by
-
Cancer immunotherapy and nanomedicine.Pharm Res. 2011 Feb;28(2):200-14. doi: 10.1007/s11095-010-0258-8. Epub 2010 Sep 4. Pharm Res. 2011. PMID: 20821040 Review.
-
Kinesin family member 11 contributes to the progression and prognosis of human breast cancer.Oncol Lett. 2017 Dec;14(6):6618-6626. doi: 10.3892/ol.2017.7053. Epub 2017 Sep 25. Oncol Lett. 2017. PMID: 29181100 Free PMC article.
-
ML323 suppresses the progression of ovarian cancer via regulating USP1-mediated cell cycle.Front Genet. 2022 Jul 18;13:917481. doi: 10.3389/fgene.2022.917481. eCollection 2022. Front Genet. 2022. PMID: 35923700 Free PMC article.
-
Ovarian cancer stem cells and inflammation.Cancer Biol Ther. 2011 Apr 15;11(8):708-13. doi: 10.4161/cbt.11.8.14967. Epub 2011 Apr 15. Cancer Biol Ther. 2011. PMID: 21317559 Free PMC article. Review.
-
Development of Half-Sandwich Ru, Os, Rh, and Ir Complexes Bearing the Pyridine-2-ylmethanimine Bidentate Ligand Derived from 7-Chloroquinazolin-4(3H)-one with Enhanced Antiproliferative Activity.ACS Omega. 2024 Apr 13;9(16):18224-18237. doi: 10.1021/acsomega.3c10482. eCollection 2024 Apr 23. ACS Omega. 2024. PMID: 38680348 Free PMC article.
References
-
- Schwartz PE. Current diagnosis and treatment modalities for ovarian cancer. Cancer Treat Res. 2002;107:99–118. - PubMed
-
- Berchuck A, Elbendary A, Havrilesky L, Rodriguez GC, Bast RC., Jr Pathogenesis of ovarian cancers. J Soc Gynecol Investig. 1994;1:181–190. - PubMed
-
- Mutch DG. Surgical management of ovarian cancer. Semin Oncol. 2002;29:3–8. - PubMed
-
- Kelly MG, Alvero AB, Chen R, Silasi DA, Abrahams VM, Chan S, Visintin I, Rutherford T, Mor G. TLR-4 signaling promotes tumor growth and paclitaxel chemoresistance in ovarian cancer. Cancer Res. 2006;66:3859–3868. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

