Dynamics of spectro-temporal tuning in primary auditory cortex of the awake ferret
- PMID: 19619629
- PMCID: PMC2808190
- DOI: 10.1016/j.heares.2009.07.005
Dynamics of spectro-temporal tuning in primary auditory cortex of the awake ferret
Abstract
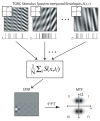
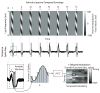
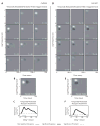
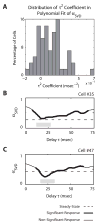
We previously characterized the steady-state spectro-temporal tuning properties of cortical cells with respect to broadband sounds by using sounds with sinusoidal spectro-temporal modulation envelope where spectral density and temporal periodicity were constant over several seconds. However, since speech and other natural sounds have spectro-temporal features that change substantially over milliseconds, we study the dynamics of tuning by using stimuli of constant overall intensity, but alternating between a flat spectro-temporal envelope and a modulated envelope with well defined spectral density and temporal periodicity. This allows us to define the tuning of cortical cells to speech-like and other rapid transitions, on the order of milliseconds, as well as the time evolution of this tuning in response to the appearance of new features in a sound. Responses of 92 cells in AI were analyzed based on the temporal evolution of the following measures of tuning after a rapid transition in the stimulus: center of mass and breadth of tuning; separability and direction selectivity; temporal and spectral asymmetry. We find that tuning center of mass increased in 70% of cells for spectral density and in 68% of cells for temporal periodicity, while roughly half of cells (47%) broadened their tuning, with the other half (53%) sharpening tuning. The majority of cells (73%) were initially not direction selective, as measured by an inseparability index, which had an initial low value that then increased to a higher steady state value. Most cells were characterized by temporal symmetry, while spectral symmetry was initially high and then progressed to low steady-state values (61%). We demonstrate that cortical neurons can be characterized by a lag-dependent modulation transfer function. This characterization, when measured through to steady-state, becomes equivalent to the classical spectro-temporal receptive field.
2009 Elsevier B.V.
Figures






Similar articles
-
Dynamics of phase-independent spectro-temporal tuning in primary auditory cortex of the awake ferret.Neuroscience. 2012 Jul 12;214:28-35. doi: 10.1016/j.neuroscience.2012.04.029. Epub 2012 Apr 21. Neuroscience. 2012. PMID: 22531376 Free PMC article.
-
Contrast tuned responses in primary auditory cortex of the awake ferret.Eur J Neurosci. 2012 Feb;35(4):550-61. doi: 10.1111/j.1460-9568.2011.07985.x. Epub 2012 Feb 9. Eur J Neurosci. 2012. PMID: 22321018
-
Stability of spectro-temporal tuning over several seconds in primary auditory cortex of the awake ferret.Neuroscience. 2007 Sep 7;148(3):806-14. doi: 10.1016/j.neuroscience.2007.06.027. Epub 2007 Aug 10. Neuroscience. 2007. PMID: 17693032 Free PMC article.
-
How do auditory cortex neurons represent communication sounds?Hear Res. 2013 Nov;305:102-12. doi: 10.1016/j.heares.2013.03.011. Epub 2013 Apr 17. Hear Res. 2013. PMID: 23603138 Review.
-
Representation of temporal sound features in the human auditory cortex.Rev Neurosci. 2011;22(2):187-203. doi: 10.1515/RNS.2011.016. Rev Neurosci. 2011. PMID: 21476940 Review.
Cited by
-
Lagged cells in the inferior colliculus of the awake ferret.Eur J Neurosci. 2010 Jan;31(1):42-8. doi: 10.1111/j.1460-9568.2009.07037.x. Epub 2009 Dec 18. Eur J Neurosci. 2010. PMID: 20092554 Free PMC article.
-
Nonlinearity of coding in primary auditory cortex of the awake ferret.Neuroscience. 2010 Jan 20;165(2):612-20. doi: 10.1016/j.neuroscience.2009.10.034. Neuroscience. 2010. PMID: 19853021 Free PMC article.
-
Dynamics of phase-independent spectro-temporal tuning in primary auditory cortex of the awake ferret.Neuroscience. 2012 Jul 12;214:28-35. doi: 10.1016/j.neuroscience.2012.04.029. Epub 2012 Apr 21. Neuroscience. 2012. PMID: 22531376 Free PMC article.
-
Auditory abstraction from spectro-temporal features to coding auditory entities.Proc Natl Acad Sci U S A. 2012 Nov 13;109(46):18968-73. doi: 10.1073/pnas.1111242109. Epub 2012 Oct 29. Proc Natl Acad Sci U S A. 2012. PMID: 23112145 Free PMC article.
References
-
- Abdi H. Singular Value Decomposition (SVD) and Generalized Singular Value Decomposition (GSVD) In: Salkind NJ, editor. Encyclopedia of Measurement and Statistics. Thousand Oaks, CA: Sage; 2007.
-
- Aertsen AM, Johannesma PI. A comparison of the spectro-temporal sensitivity of auditory neurons to tonal and natural stimuli. Biological Cybernetics. 1981;42:145–156. - PubMed
-
- deCharms RC, Blake DT, Merzenich MM. Optimizing sound features for cortical neurons.[see comment] Science. 1998;280:1439–1443. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

