Calcium-activated chloride currents in olfactory sensory neurons from mice lacking bestrophin-2
- PMID: 19622610
- PMCID: PMC2754364
- DOI: 10.1113/jphysiol.2009.176131
Calcium-activated chloride currents in olfactory sensory neurons from mice lacking bestrophin-2
Abstract
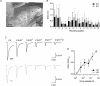
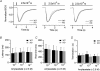
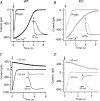
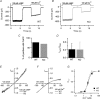
Olfactory sensory neurons use a chloride-based signal amplification mechanism to detect odorants. The binding of odorants to receptors in the cilia of olfactory sensory neurons activates a transduction cascade that involves the opening of cyclic nucleotide-gated channels and the entry of Ca(2+) into the cilia. Ca(2+) activates a Cl(-) current that produces an efflux of Cl(-) ions and amplifies the depolarization. The molecular identity of Ca(2+)-activated Cl(-) channels is still elusive, although some bestrophins have been shown to function as Ca(2+)-activated Cl(-) channels when expressed in heterologous systems. In the olfactory epithelium, bestrophin-2 (Best2) has been indicated as a candidate for being a molecular component of the olfactory Ca(2+)-activated Cl(-) channel. In this study, we have analysed mice lacking Best2. We compared the electrophysiological responses of the olfactory epithelium to odorant stimulation, as well as the properties of Ca(2+)-activated Cl(-) currents in wild-type (WT) and knockout (KO) mice for Best2. Our results confirm that Best2 is expressed in the cilia of olfactory sensory neurons, while odorant responses and Ca(2+)-activated Cl(-) currents were not significantly different between WT and KO mice. Thus, Best2 does not appear to be the main molecular component of the olfactory channel. Further studies are required to determine the function of Best2 in the cilia of olfactory sensory neurons.
Figures


Comment in
-
Identifying olfaction's 'other channels'.J Physiol. 2009 Sep 1;587(Pt 17):4135-6. doi: 10.1113/jphysiol.2009.178954. J Physiol. 2009. PMID: 19720851 Free PMC article. No abstract available.
Similar articles
-
Bestrophin-2 is a candidate calcium-activated chloride channel involved in olfactory transduction.Proc Natl Acad Sci U S A. 2006 Aug 22;103(34):12929-34. doi: 10.1073/pnas.0604505103. Epub 2006 Aug 15. Proc Natl Acad Sci U S A. 2006. PMID: 16912113 Free PMC article.
-
Single Ca(2+)-activated Cl(-) channel currents recorded from toad olfactory cilia.BMC Neurosci. 2016 Apr 25;17(1):17. doi: 10.1186/s12868-016-0252-0. BMC Neurosci. 2016. PMID: 27113933 Free PMC article.
-
Anoctamin 2/TMEM16B: a calcium-activated chloride channel in olfactory transduction.Exp Physiol. 2012 Feb;97(2):193-9. doi: 10.1113/expphysiol.2011.058230. Epub 2011 Sep 2. Exp Physiol. 2012. PMID: 21890523 Review.
-
Mechanism of olfactory masking in the sensory cilia.J Gen Physiol. 2009 Jun;133(6):583-601. doi: 10.1085/jgp.200810085. Epub 2009 May 11. J Gen Physiol. 2009. PMID: 19433623 Free PMC article.
-
Bestrophin and TMEM16-Ca(2+) activated Cl(-) channels with different functions.Cell Calcium. 2009 Oct;46(4):233-41. doi: 10.1016/j.ceca.2009.09.003. Epub 2009 Sep 26. Cell Calcium. 2009. PMID: 19783045 Review.
Cited by
-
Deep sequencing of the murine olfactory receptor neuron transcriptome.PLoS One. 2015 Jan 15;10(1):e0113170. doi: 10.1371/journal.pone.0113170. eCollection 2015. PLoS One. 2015. PMID: 25590618 Free PMC article.
-
Functional expression of TMEM16A in taste bud cells.J Physiol. 2021 Aug;599(15):3697-3714. doi: 10.1113/JP281645. Epub 2021 Jun 28. J Physiol. 2021. PMID: 34089532 Free PMC article.
-
Calcium-activated chloride channels in the apical region of mouse vomeronasal sensory neurons.J Gen Physiol. 2012 Jul;140(1):3-15. doi: 10.1085/jgp.201210780. J Gen Physiol. 2012. PMID: 22732308 Free PMC article.
-
Signal Detection and Coding in the Accessory Olfactory System.Chem Senses. 2018 Nov 1;43(9):667-695. doi: 10.1093/chemse/bjy061. Chem Senses. 2018. PMID: 30256909 Free PMC article. Review.
-
Calcium concentration jumps reveal dynamic ion selectivity of calcium-activated chloride currents in mouse olfactory sensory neurons and TMEM16b-transfected HEK 293T cells.J Physiol. 2010 Nov 1;588(Pt 21):4189-204. doi: 10.1113/jphysiol.2010.194407. Epub 2010 Sep 13. J Physiol. 2010. PMID: 20837642 Free PMC article.
References
-
- Boccaccio A, Menini A. Temporal development of cyclic nucleotide-gated and Ca2+-activated Cl– currents in isolated mouse olfactory sensory neurons. J Neurophysiol. 2007;98:153–160. - PubMed
-
- Caputo A, Caci E, Ferrera L, Pedemonte N, Barsanti C, Sondo E, Pfeffer U, Ravazzolo R, Zegarra-Moran O, Galietta LJ. TMEM16A, a membrane protein associated with calcium-dependent chloride channel activity. Science. 2008;322:590–594. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

