Intramitochondrial calcium regulation by the FHIT gene product sensitizes to apoptosis
- PMID: 19622739
- PMCID: PMC2722368
- DOI: 10.1073/pnas.0906484106
Intramitochondrial calcium regulation by the FHIT gene product sensitizes to apoptosis
Abstract
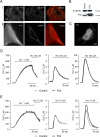
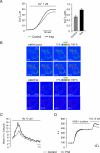
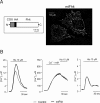
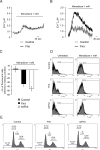
Despite the growing interest in the Fhit tumor suppressor protein, frequently deleted in human cancers, the mechanism of its powerful proapoptotic activity has remained elusive. We here demonstrate that Fhit sensitizes the low-affinity Ca(2+) transporters of mitochondria, enhancing Ca(2+) uptake into the organelle both in intact and in permabilized cells, and potentiating the effect of apoptotic agents. This effect can be attributed to the fraction of Fhit sorted to mitochondria, as a fully mitochondrial Fhit (a chimeric protein including a mitochondrial targeting sequence) retains the Ca(2+) signaling properties of Fhit and the proapoptotic activity of the native protein (whereas the effects on the cell cycle are lost). Thus, the partial sorting of Fhit to mitochondria allows to finely tune the sensitivity of the organelle to the highly pleiomorphic Ca(2+) signals, synergizing with apoptotic challenges. This concept, and the identification of the molecular machinery, may provide ways to act on apoptotic cell death and its derangement in cancer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
The apoptotic pathway triggered by the Fhit protein in lung cancer cell lines is not affected by Bcl-2 or Bcl-x(L) overexpression.Oncogene. 2004 Dec 2;23(56):9102-10. doi: 10.1038/sj.onc.1208142. Oncogene. 2004. PMID: 15489891
-
[FHIT--tumor suppressor protein involved in induction of apoptosis and cell cycle regulation].Postepy Biochem. 2009;55(1):66-75. Postepy Biochem. 2009. PMID: 19514467 Review. Polish.
-
Implication of mitochondrial involvement in apoptotic activity of fragile histidine triad gene: application of synchronous luminescence spectroscopy.Biopolymers. 2004 Mar;73(4):510-23. doi: 10.1002/bip.10544. Biopolymers. 2004. PMID: 14991669
-
Aberration of the enzymatic activity of Fhit tumor suppressor protein enhances cancer cell death upon photodynamic therapy similarly to that driven by wild-type Fhit.Cancer Lett. 2009 Jul 18;280(1):101-9. doi: 10.1016/j.canlet.2009.02.014. Epub 2009 Mar 14. Cancer Lett. 2009. PMID: 19286306
-
Enlightened protein: Fhit tumor suppressor protein structure and function and its role in the toxicity of protoporphyrin IX-mediated photodynamic reaction.Toxicol Appl Pharmacol. 2009 Dec 1;241(2):246-52. doi: 10.1016/j.taap.2009.08.023. Epub 2009 Aug 28. Toxicol Appl Pharmacol. 2009. PMID: 19716840 Review.
Cited by
-
The effects of Fhit on tumorigenesis after multi-exposure to low-dose radiation.Int J Clin Exp Med. 2009 Nov 22;2(4):348-53. Int J Clin Exp Med. 2009. PMID: 20057978 Free PMC article.
-
The clinicopathological significance and drug target potential of FHIT in breast cancer, a meta-analysis and literature review.Drug Des Devel Ther. 2015 Oct 1;9:5439-45. doi: 10.2147/DDDT.S89861. eCollection 2015. Drug Des Devel Ther. 2015. PMID: 26491255 Free PMC article. Review.
-
Endoplasmic Reticulum-Mitochondrial Ca2+ Fluxes Underlying Cancer Cell Survival.Front Oncol. 2017 May 3;7:70. doi: 10.3389/fonc.2017.00070. eCollection 2017. Front Oncol. 2017. PMID: 28516062 Free PMC article. Review.
-
Fhit delocalizes annexin a4 from plasma membrane to cytosol and sensitizes lung cancer cells to paclitaxel.PLoS One. 2013 Nov 6;8(11):e78610. doi: 10.1371/journal.pone.0078610. eCollection 2013. PLoS One. 2013. PMID: 24223161 Free PMC article.
-
Mitochondria associated membranes (MAMs) as critical hubs for apoptosis.Commun Integr Biol. 2011 May;4(3):334-5. doi: 10.4161/cib.4.3.15021. Commun Integr Biol. 2011. PMID: 21980573 Free PMC article.
References
-
- Huebner K, Croce CM. FRA3B and other common fragile sites: The weakest links. Nat Rev Cancer. 2001;1:214–221. - PubMed
-
- Ohta M, et al. The FHIT gene, spanning the chromosome 3p14.2 fragile site and renal carcinoma-associated t(3;8) breakpoint, is abnormal in digestive tract cancers. Cell. 1996;84:587–597. - PubMed
-
- Toledo G, Sola JJ, Lozano MD, Soria E, Pardo J. Loss of FHIT protein expression is related to high proliferation, low apoptosis and worse prognosis in non-small-cell lung cancer. Mod Pathol. 2004;17:440–448. - PubMed
-
- Pekarsky Y, Palamarchuk A, Huebner K, Croce CM. FHIT as tumor suppressor: Mechanisms and therapeutic opportunities. Cancer Biol Ther. 2002;1:232–236. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

