Risedronate inhibits human osteosarcoma cell invasion
- PMID: 19624845
- PMCID: PMC2729740
- DOI: 10.1186/1756-9966-28-105
Risedronate inhibits human osteosarcoma cell invasion
Abstract
Background: Osteosarcoma is a highly malignant bone tumor and is the most commonly encountered malignant bone tumor in children and adolescents. Furthermore, significant numbers of patients eventually develop pulmonary metastases and succumb to the disease even after conventional multi-agent chemotherapy and surgical excision. Several solid tumors display enhanced expression of matrix metalloproteinases (MMPs), and recently clinical trials have been initiated on MMP-inhibitors. On the other hand, bisphosphonates (BPs), which have a profound effect on bone resorption, are widely used to treat osteoclast-mediated bone diseases. BPs are also known to inhibit tumor growths and metastases in some tumors such as breast cancer, renal cell carcinoma, and prostate cancer.
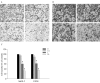
Methods: Two osteosarcoma cell lines (SaOS-2 and U2OS) were treated with risedronate (0, 0.1, 1, 10 microM) for 48 hours. Cell viabilities were determined using MTT assay, the mRNA levels of MMP-2 and MMP-9 were analyzed by reverse-transcription polymerase chain reaction, the amount of MMP-2 and MMP-9 protein were analyzed by Westernblot, the activities of MMP-2 and MMP-9 were observed by Gelatin zymography, and Matrigel invasion assays were used to investigate the invasive potential of osteosarcoma cell lines before and after risedronate treatment.
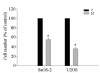
Results: The invasiveness of osteosarcoma cell lines (SaOS-2, U2OS) were reduced in a dose dependent manner follow 48 hour treatment of up to 10 microM of the risedronate at which concentration no cytotoxicity occurred. Furthermore, the gelatinolytic activities and protein and mRNA levels of MMP-2 and MMP-9 were also suppressed by increasing risedronate concentrations.
Conclusion: Given that MMP-2 and MMP-9 are instrumental in tumor cell invasion, our results suggest the risedronate could reduce osteosarcoma cell invasion.
Figures
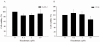



References
-
- Hauben EI, Arends J, Vandenbroucke JP, van Asperen CJ, Van Marck E, Hogendoorn PC. Multiple primary malignancies in osteosarcoma patients. Incidence and predictive value of osteosarcoma subtype for cancer syndromes related with osteosarcoma. Eur J Human Genetics. 2003;11:611–618. doi: 10.1038/sj.ejhg.5201012. - DOI - PubMed
-
- Link MP. In: Preoperative and adjuvant chemotherapy in osteosarcoma. In Frontiers of Osteosarcoma Research: Interdisciplinary Survey of Clinical and Research Advances. Novak JF, Mcmaster JH, editor. Seattle: Hogrefe and Huber; 1993. pp. 41–49.
-
- Unni KK. Dahlin' BONE TUMORS General Aspects and Date on 11, 0809 Cases. 5. Philadelphia: Lippincott; 1996. pp. 143–183.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

