Antibody limits in vivo murid herpesvirus-4 replication by IgG Fc receptor-dependent functions
- PMID: 19625459
- PMCID: PMC2885036
- DOI: 10.1099/vir.0.014266-0
Antibody limits in vivo murid herpesvirus-4 replication by IgG Fc receptor-dependent functions
Abstract
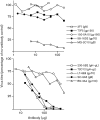
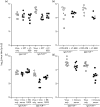
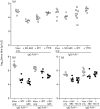
Antibody is an important antiviral defence. However, it is considered to do little against human gamma-herpesviruses, which establish predominantly latent infections regulated by T cells. One limitation on analysing these infections has been that latency is already well-established at clinical presentation; early infection may still be accessible to antibody. Here, using murid herpesvirus-4 (MuHV-4), we tested the impact of adoptively transferred antibody on early gamma-herpesvirus infection. Immune sera and neutralizing and non-neutralizing monoclonal antibodies (mAbs) all reduced acute lytic MuHV-4 replication. The reductions, even by neutralizing mAbs, were largely or completely dependent on host IgG Fc receptors. Therefore, passive antibody can blunt acute gamma-herpesvirus lytic infection, and does this principally by IgG Fc-dependent functions rather than by neutralization.
Figures




Similar articles
-
Vaccination with murid herpesvirus-4 glycoprotein B reduces viral lytic replication but does not induce detectable virion neutralization.J Gen Virol. 2010 Oct;91(Pt 10):2542-52. doi: 10.1099/vir.0.023085-0. Epub 2010 Jun 2. J Gen Virol. 2010. PMID: 20519454 Free PMC article.
-
IgG fc receptors provide an alternative infection route for murine gamma-herpesvirus-68.PLoS One. 2007 Jun 27;2(6):e560. doi: 10.1371/journal.pone.0000560. PLoS One. 2007. PMID: 17593961 Free PMC article.
-
The murine gammaherpesvirus-68 gp150 acts as an immunogenic decoy to limit virion neutralization.PLoS One. 2007 Aug 8;2(8):e705. doi: 10.1371/journal.pone.0000705. PLoS One. 2007. PMID: 17684552 Free PMC article.
-
Immune control of mammalian gamma-herpesviruses: lessons from murid herpesvirus-4.J Gen Virol. 2009 Oct;90(Pt 10):2317-2330. doi: 10.1099/vir.0.013300-0. Epub 2009 Jul 15. J Gen Virol. 2009. PMID: 19605591 Review.
-
Fc-Mediated Antibody Effector Functions During Respiratory Syncytial Virus Infection and Disease.Front Immunol. 2019 Mar 22;10:548. doi: 10.3389/fimmu.2019.00548. eCollection 2019. Front Immunol. 2019. PMID: 30967872 Free PMC article. Review.
Cited by
-
Vaccination with murid herpesvirus-4 glycoprotein B reduces viral lytic replication but does not induce detectable virion neutralization.J Gen Virol. 2010 Oct;91(Pt 10):2542-52. doi: 10.1099/vir.0.023085-0. Epub 2010 Jun 2. J Gen Virol. 2010. PMID: 20519454 Free PMC article.
-
B cell response to herpesvirus infection of the olfactory neuroepithelium.J Virol. 2014 Dec;88(24):14030-9. doi: 10.1128/JVI.02345-14. Epub 2014 Sep 24. J Virol. 2014. PMID: 25253348 Free PMC article.
-
Indirect CD4+ T cell protection against mouse gamma-herpesvirus infection via interferon gamma.J Virol. 2024 May 14;98(5):e0049324. doi: 10.1128/jvi.00493-24. Epub 2024 Apr 5. J Virol. 2024. PMID: 38578092 Free PMC article.
-
Caspase cleavage of influenza A virus M2 disrupts M2-LC3 interaction and regulates virion production.EMBO Rep. 2025 Apr;26(7):1768-1791. doi: 10.1038/s44319-025-00388-7. Epub 2025 Mar 3. EMBO Rep. 2025. PMID: 40033051 Free PMC article.
-
A CD4+ T Cell-NK Cell Axis of Gammaherpesvirus Control.J Virol. 2020 Jan 17;94(3):e01545-19. doi: 10.1128/JVI.01545-19. Print 2020 Jan 17. J Virol. 2020. PMID: 31694958 Free PMC article.
References
-
- Callan, M. F., Steven, N., Krausa, P., Wilson, J. D., Moss, P. A., Gillespie, G. M., Bell, J. I., Rickinson, A. B. & McMichael, A. J. (1996). Large clonal expansions of CD8+ T cells in acute infectious mononucleosis. Nat Med 2, 906–911. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

