Alternative splicing in the voltage-gated sodium channel DmNav regulates activation, inactivation, and persistent current
- PMID: 19625535
- PMCID: PMC2746785
- DOI: 10.1152/jn.00613.2009
Alternative splicing in the voltage-gated sodium channel DmNav regulates activation, inactivation, and persistent current
Abstract
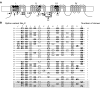
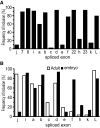
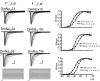
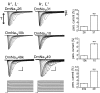
Diversity in neuronal signaling is a product not only of differential gene expression, but also of alternative splicing. However, although recognized, the precise contribution of alternative splicing in ion channel transcripts to channel kinetics remains poorly understood. Invertebrates, with their smaller genomes, offer attractive models to examine the contribution of splicing to neuronal function. In this study we report the sequencing and biophysical characterization of alternative splice variants of the sole voltage-gated Na+ gene (DmNav, paralytic), in late-stage embryos of Drosophila melanogaster. We identify 27 unique splice variants, based on the presence of 15 alternative exons. Heterologous expression, in Xenopus oocytes, shows that alternative exons j, e, and f primarily influence activation kinetics: when present, exon f confers a hyperpolarizing shift in half-activation voltage (V1/2), whereas j and e result in a depolarizing shift. The presence of exon h is sufficient to produce a depolarizing shift in the V1/2 of steady-state inactivation. The magnitude of the persistent Na+ current, but not the fast-inactivating current, in both oocytes and Drosophila motoneurons in vivo is directly influenced by the presence of either one of a pair of mutually exclusive, membrane-spanning exons, termed k and L. Transcripts containing k have significantly smaller persistent currents compared with those containing L. Finally, we show that transcripts lacking all cytoplasmic alternatively spliced exons still produce functional channels, indicating that splicing may influence channel kinetics not only through change to protein structure, but also by allowing differential modification (i.e., phosphorylation, binding of cofactors, etc.). Our results provide a functional basis for understanding how alternative splicing of a voltage-gated Na+ channel results in diversity in neuronal signaling.
Figures



Similar articles
-
Alternative splicing of an insect sodium channel gene generates pharmacologically distinct sodium channels.J Neurosci. 2002 Jul 1;22(13):5300-9. doi: 10.1523/JNEUROSCI.22-13-05300.2002. J Neurosci. 2002. PMID: 12097481 Free PMC article.
-
Activity-dependent alternative splicing increases persistent sodium current and promotes seizure.J Neurosci. 2012 May 23;32(21):7267-77. doi: 10.1523/JNEUROSCI.6042-11.2012. J Neurosci. 2012. PMID: 22623672 Free PMC article.
-
A novel Na+ channel splice form contributes to the regulation of an androgen-dependent social signal.J Neurosci. 2008 Sep 10;28(37):9173-82. doi: 10.1523/JNEUROSCI.2783-08.2008. J Neurosci. 2008. PMID: 18784298 Free PMC article.
-
Functional regulation of alternatively spliced Na+/Ca2+ exchanger (NCX1) isoforms.Ann N Y Acad Sci. 2002 Nov;976:187-96. doi: 10.1111/j.1749-6632.2002.tb04740.x. Ann N Y Acad Sci. 2002. PMID: 12502560 Review.
-
The molecular interactions of pyrethroid insecticides with insect and mammalian sodium channels.Pest Manag Sci. 2001 Oct;57(10):877-88. doi: 10.1002/ps.392. Pest Manag Sci. 2001. PMID: 11695180 Review.
Cited by
-
Membranes with the same ion channel populations but different excitabilities.PLoS One. 2012;7(4):e34636. doi: 10.1371/journal.pone.0034636. Epub 2012 Apr 16. PLoS One. 2012. PMID: 22523552 Free PMC article.
-
A knock-in model of human epilepsy in Drosophila reveals a novel cellular mechanism associated with heat-induced seizure.J Neurosci. 2012 Oct 10;32(41):14145-55. doi: 10.1523/JNEUROSCI.2932-12.2012. J Neurosci. 2012. PMID: 23055484 Free PMC article.
-
Chemical and Biological Tools for the Study of Voltage-Gated Sodium Channels in Electrogenesis and Nociception.Chembiochem. 2022 Jul 5;23(13):e202100625. doi: 10.1002/cbic.202100625. Epub 2022 Mar 21. Chembiochem. 2022. PMID: 35315190 Free PMC article. Review.
-
Dual separable feedback systems govern firing rate homeostasis.Elife. 2019 Apr 11;8:e45717. doi: 10.7554/eLife.45717. Elife. 2019. PMID: 30973325 Free PMC article.
-
Development of modified cable models to simulate accurate neuronal active behaviors.J Appl Physiol (1985). 2014 Dec 1;117(11):1243-61. doi: 10.1152/japplphysiol.00496.2014. Epub 2014 Oct 2. J Appl Physiol (1985). 2014. PMID: 25277743 Free PMC article.
References
-
- Auld VJ, Goldin AL, Krafte DS, Marshall J, Dunn JM, Catterall WA, Lester HA, Davidson N, Dunn RJ. A rat brain Na+ channel alpha subunit with novel gating properties. Neuron 1: 449–461, 1988. - PubMed
-
- Brose K, Tessier-Lavigne M. Slit proteins: key regulators of axon guidance, axonal branching, and cell migration. Curr Opin Neurobiol 10: 95–102, 2000. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

