A non-apoptotic function of caspase-3 in pharmacologically-induced differentiation of K562 cells
- PMID: 19627286
- PMCID: PMC2765302
- DOI: 10.1111/j.1476-5381.2009.00333.x
A non-apoptotic function of caspase-3 in pharmacologically-induced differentiation of K562 cells
Abstract
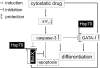
Background and purpose: Several anticancer drugs with diverse chemical structures can induce differentiation of cancer cells. This study was undertaken to explore the potential contribution of caspase-3 to pharmacologically-induced differentiation of K562 cells.
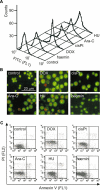
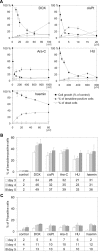
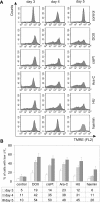
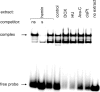
Experimental approach: We assessed differentiation by measuring the expression of glycophorin A and haemoglobin synthesis in K562 cells treated with low concentrations of doxorubicin, hydroxyurea, cytosine arabinoside, cisplatin and haemin. Caspase-3 activation, mitochondrial membrane potential dissipation and viability were assessed by FACS. GATA-1-binding activity was evaluated by EMSA.
Key results: Treatment of K562 cells with low concentrations of the tested drugs activated caspase-3 but did not trigger detectable apoptosis. Instead, elevated levels of haemoglobin-positive and glycophorin A/caspase-3-double-positive cells were observed, suggesting involvement of caspase-3 in drug-induced differentiation. Inhibition of caspase-3 activity significantly reduced the ability of K562 cells to execute the differentiation programme. Mitochondrial membrane potential dissipation was observed, indicating involvement of the mitochondrial pathway. Binding activity of GATA-1, transcription factor responsible for differentiation and cell survival, was not diminished by increased caspase-3 activity during drug-stimulated differentiation.
Conclusions and implications: Our results could explain how anticancer drugs, with diverse structures and modes of action, can stimulate erythroid differentiation in leukaemic cells with appropriate genetic backgrounds. Our findings imply that some similarities exist between pharmacologically-induced differentiation of erythroleukaemic cells and normal erythropoiesis, both involving caspase-3 activation at high levels of anti-apoptotic protein Bcl-X(L) and chaperone protein Hsp70 (heat shock protein 70). Therefore, the functions of caspase-3, unrelated to cell death, can be extended to pharmacologically-induced differentiation of some cancer cells.
Figures

Similar articles
-
Hsp70 regulates erythropoiesis by preventing caspase-3-mediated cleavage of GATA-1.Nature. 2007 Jan 4;445(7123):102-5. doi: 10.1038/nature05378. Epub 2006 Dec 10. Nature. 2007. PMID: 17167422
-
Induction of apoptosis and erythroid differentiation of human chronic myelogenous leukemia K562 cells by low concentrations of lidamycin.Oncol Rep. 2019 Jan;41(1):475-482. doi: 10.3892/or.2018.6849. Epub 2018 Nov 2. Oncol Rep. 2019. PMID: 30542729
-
STI571 and morpholine derivative of doxorubicin collaborate in inhibition of K562 cell proliferation by inducing differentiation and mitochondrial pathway of apoptosis.Eur J Pharmacol. 2008 Oct 31;596(1-3):41-9. doi: 10.1016/j.ejphar.2008.08.021. Epub 2008 Aug 30. Eur J Pharmacol. 2008. PMID: 18782571
-
[HSP70, an erythropoiesis regulator that determines the fate of erythroblasts between death and differentiation].Transfus Clin Biol. 2013 May;20(2):144-7. doi: 10.1016/j.tracli.2013.03.005. Epub 2013 May 2. Transfus Clin Biol. 2013. PMID: 23643330 Review. French.
-
[Erythropoiesis: a paradigm for the role of caspases in cell death and differentiation].J Soc Biol. 2005;199(3):219-31. doi: 10.1051/jbio:2005023. J Soc Biol. 2005. PMID: 16471262 Review. French.
Cited by
-
Chemoprotective effect of atorvastatin against benzo(a)pyrene-induced lung cancer via the inhibition of oxidative stress and inflammatory parameters.Ann Transl Med. 2021 Feb;9(4):355. doi: 10.21037/atm-20-7770. Ann Transl Med. 2021. PMID: 33708982 Free PMC article.
-
Mapping enzyme activity in living systems by real-time mid-infrared photothermal imaging of nitrile chameleons.Nat Methods. 2024 Feb;21(2):342-352. doi: 10.1038/s41592-023-02137-x. Epub 2024 Jan 8. Nat Methods. 2024. PMID: 38191931 Free PMC article.
-
Estrogen receptor-alpha 36 mediates the anti-apoptotic effect of estradiol in triple negative breast cancer cells via a membrane-associated mechanism.Biochim Biophys Acta. 2014 Nov;1843(11):2796-806. doi: 10.1016/j.bbamcr.2014.07.019. Epub 2014 Aug 7. Biochim Biophys Acta. 2014. PMID: 25108195 Free PMC article.
-
Development of high content imaging methods for cell death detection in human pluripotent stem cell-derived cardiomyocytes.J Cardiovasc Transl Res. 2012 Oct;5(5):593-604. doi: 10.1007/s12265-012-9396-1. Epub 2012 Aug 16. J Cardiovasc Transl Res. 2012. PMID: 22896035 Free PMC article.
-
Caspase-3 cleaves hnRNP K in erythroid differentiation.Cell Death Dis. 2013 Mar 21;4(3):e548. doi: 10.1038/cddis.2013.75. Cell Death Dis. 2013. PMID: 23519117 Free PMC article.
References
-
- Addya S, Keller MA, Delgrosso K, Ponte CM, Vadigepalli R, Gonye GE, et al. Erythroid-induced commitment of K562 cells results in clusters of differentially expressed genes enriched for specific transcription regulatory elements. Physiol Genomics. 2004;19:117–130. - PubMed
-
- Aispuru GR, Aguirre MV, Aquino-Esperanza JA, Lettieri CN, Juaristi JA, Brandan NC. Erythroid expansion and survival in response to acute anemia stress: the role of EPO receptor, GATA-1, Bcl-xL and caspase-3. Cell Biol Int. 2008;32:966–978. - PubMed
-
- Baliga BS, Mankad M, Shah AK, Mankad VN. Mechanism of differentiation of human erythroleukaemic cell line K562 by haemin. Cell Prolif. 1993;26:519–529. - PubMed
-
- Belhacène N, Maulon L, Guérin S, Ricci JE, Mari B, Colin Y, et al. Differential expression of the Kell blood group and CD10 antigens: two related membrane metallopeptidases during differentiation of K562 cells by phorbol ester and hemin. FASEB J. 1998;12:531–539. - PubMed
-
- Benito A, Silva M, Grillot D, Nuñez G, Fernández-Luna JL. Apoptosis induced by erythroid differentiation of human leukemia cell lines is inhibited by Bcl-XL. Blood. 1996;87:3837–3843. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

