The use of antibiotics to improve phage detection and enumeration by the double-layer agar technique
- PMID: 19627589
- PMCID: PMC2728735
- DOI: 10.1186/1471-2180-9-148
The use of antibiotics to improve phage detection and enumeration by the double-layer agar technique
Abstract
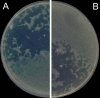
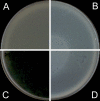
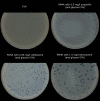
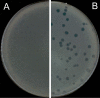
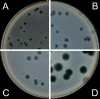
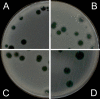
Background: The Double-Layer Agar (DLA) technique is extensively used in phage research to enumerate and identify phages and to isolate mutants and new phages. Many phages form large and well-defined plaques that are easily observed so that they can be enumerated when plated by the DLA technique. However, some give rise to small and turbid plaques that are very difficult to detect and count. To overcome these problems, some authors have suggested the use of dyes to improve the contrast between the plaques and the turbid host lawns. It has been reported that some antibiotics stimulate bacteria to produce phages, resulting in an increase in final titer. Thus, antibiotics might contribute to increasing plaque size in solid media.
Results: Antibiotics with different mechanisms of action were tested for their ability to enhance plaque morphology without suppressing phage development. Some antibiotics increased the phage plaque surface by up to 50-fold.
Conclusion: This work presents a modification of the DLA technique that can be used routinely in the laboratory, leading to a more accurate enumeration of phages that would be difficult or even impossible otherwise.
Figures




Similar articles
-
Simple drop cast method for enumeration of bacteriophages.J Virol Methods. 2018 Dec;262:1-5. doi: 10.1016/j.jviromet.2018.09.001. Epub 2018 Sep 10. J Virol Methods. 2018. PMID: 30213546
-
Methicillin-resistant Staphylococcus aureus phage plaque size enhancement using sublethal concentrations of antibiotics.Appl Environ Microbiol. 2012 Dec;78(23):8227-33. doi: 10.1128/AEM.02371-12. Epub 2012 Sep 21. Appl Environ Microbiol. 2012. PMID: 23001655 Free PMC article.
-
A single-layer spot assay for easy, fast, and high-throughput quantitation of phages against multidrug-resistant Gram-negative pathogens.J Clin Microbiol. 2024 Aug 14;62(8):e0074324. doi: 10.1128/jcm.00743-24. Epub 2024 Jul 29. J Clin Microbiol. 2024. PMID: 39072625 Free PMC article.
-
In-Gel Isolation and Characterization of Large (and Other) Phages.Viruses. 2020 Apr 7;12(4):410. doi: 10.3390/v12040410. Viruses. 2020. PMID: 32272774 Free PMC article. Review.
-
Bacteriophage inspired antibiotics discovery against infection involved biofilm.Crit Rev Eukaryot Gene Expr. 2013;23(4):317-26. doi: 10.1615/critreveukaryotgeneexpr.2013007717. Crit Rev Eukaryot Gene Expr. 2013. PMID: 24266847 Review.
Cited by
-
Endophytic fungi harbored in Panax notoginseng: diversity and potential as biological control agents against host plant pathogens of root-rot disease.J Ginseng Res. 2017 Jul;41(3):353-360. doi: 10.1016/j.jgr.2016.07.005. Epub 2016 Jul 20. J Ginseng Res. 2017. PMID: 28701877 Free PMC article.
-
Isolation and in vitro evaluation of bacteriophages against MDR-bacterial isolates from septic wound infections.PLoS One. 2017 Jul 18;12(7):e0179245. doi: 10.1371/journal.pone.0179245. eCollection 2017. PLoS One. 2017. PMID: 28719657 Free PMC article.
-
Phage-Antibiotic Therapy as a Promising Strategy to Combat Multidrug-Resistant Infections and to Enhance Antimicrobial Efficiency.Antibiotics (Basel). 2022 Apr 25;11(5):570. doi: 10.3390/antibiotics11050570. Antibiotics (Basel). 2022. PMID: 35625214 Free PMC article. Review.
-
Antibiofilm Efficacy of the Pseudomonas aeruginosa Pbunavirus vB_PaeM-SMS29 Loaded onto Dissolving Polyvinyl Alcohol Microneedles.Viruses. 2022 May 5;14(5):964. doi: 10.3390/v14050964. Viruses. 2022. PMID: 35632706 Free PMC article.
-
Isolation and Characterization of a Novel Virulent Phage ASG01 of Aeromonas salmonicida and Its Cell Wall Hydrolase Activity.Microorganisms. 2024 Mar 21;12(3):629. doi: 10.3390/microorganisms12030629. Microorganisms. 2024. PMID: 38543679 Free PMC article.
References
-
- Serwer P, Hayes SJ, Zaman S, Lieman K, Rolando M, Hardies SC. Improved isolation of undersampled bacteriophages: finding of distant terminase genes. Virology. 2004;329:412–424. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

