The pancreatic zymogen granule membrane protein, GP2, binds Escherichia coli Type 1 fimbriae
- PMID: 19627615
- PMCID: PMC2726147
- DOI: 10.1186/1471-230X-9-58
The pancreatic zymogen granule membrane protein, GP2, binds Escherichia coli Type 1 fimbriae
Abstract
Background: GP2 is the major membrane protein present in the pancreatic zymogen granule, and is cleaved and released into the pancreatic duct along with exocrine secretions. The function of GP2 is unknown. GP2's amino acid sequence is most similar to that of uromodulin, which is secreted by the kidney. Recent studies have demonstrated uromodulin binding to bacterial Type 1 fimbria. The fimbriae serve as adhesins to host receptors. The present study examines whether GP2 also shares similar binding properties to bacteria with Type 1 fimbria. Commensal and pathogenic bacteria, including E. coli and Salmonella, express type 1 fimbria.
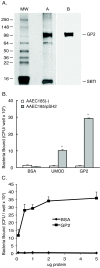
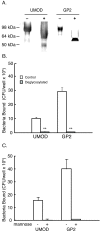
Methods: An in vitro binding assay was used to assay the binding of recombinant GP2 to defined strains of E. coli that differ in their expression of Type 1 fimbria or its subunit protein, FimH. Studies were also performed to determine whether GP2 binding is dependent on the presence of mannose residues, which is a known determinant for FimH binding.
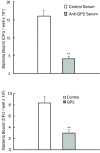
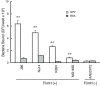
Results: GP2 binds E. coli that express Type 1 fimbria. Binding is dependent on GP2 glycosylation, and specifically the presence of mannose residues.
Conclusion: GP2 binds to Type 1 fimbria, a bacterial adhesin that is commonly expressed by members of the Enterobacteriacae family.
Figures
Similar articles
-
Adhesion of Enteropathogenic, Enterotoxigenic, and Commensal Escherichia coli to the Major Zymogen Granule Membrane Glycoprotein 2.Appl Environ Microbiol. 2022 Mar 8;88(5):e0227921. doi: 10.1128/aem.02279-21. Epub 2022 Jan 12. Appl Environ Microbiol. 2022. PMID: 35020452 Free PMC article.
-
Tamm-Horsfall protein binds to type 1 fimbriated Escherichia coli and prevents E. coli from binding to uroplakin Ia and Ib receptors.J Biol Chem. 2001 Mar 30;276(13):9924-30. doi: 10.1074/jbc.M008610200. Epub 2000 Dec 27. J Biol Chem. 2001. PMID: 11134021
-
Characterization of 20K fimbria, a new adhesin of septicemic and diarrhea-associated Escherichia coli strains, that belongs to a family of adhesins with N-acetyl-D-glucosamine recognition.Infect Immun. 1996 Jan;64(1):332-42. doi: 10.1128/iai.64.1.332-342.1996. Infect Immun. 1996. PMID: 8557360 Free PMC article.
-
Glycoprotein 2 (GP2): grabbing the FimH bacteria into M cells for mucosal immunity.Gut Microbes. 2010 Nov-Dec;1(6):407-10. doi: 10.4161/gmic.1.6.14078. Gut Microbes. 2010. PMID: 21468225 Free PMC article. Review.
-
The sweet connection: Solving the riddle of multiple sugar-binding fimbrial adhesins in Escherichia coli: Multiple E. coli fimbriae form a versatile arsenal of sugar-binding lectins potentially involved in surface-colonisation and tissue tropism.Bioessays. 2011 Apr;33(4):300-11. doi: 10.1002/bies.201000121. Epub 2011 Feb 22. Bioessays. 2011. PMID: 21344461 Review.
Cited by
-
Bacteria-Induced Dscam Isoforms of the Crustacean, Pacifastacus leniusculus.PLoS Pathog. 2011 Jun;7(6):e1002062. doi: 10.1371/journal.ppat.1002062. Epub 2011 Jun 9. PLoS Pathog. 2011. Retraction in: PLoS Pathog. 2016 May 04;12(5):e1005630. doi: 10.1371/journal.ppat.1005630. PMID: 21695245 Free PMC article. Retracted.
-
Mucosal Autoimmunity to Cell-Bound GP2 Isoforms Is a Sensitive Marker in PSC and Associated With the Clinical Phenotype.Front Immunol. 2018 Aug 28;9:1959. doi: 10.3389/fimmu.2018.01959. eCollection 2018. Front Immunol. 2018. PMID: 30233574 Free PMC article. Clinical Trial.
-
Comparison of milk fat globule membrane (MFGM) proteins of Chianina and Holstein cattle breed milk samples through proteomics methods.Nutrients. 2009 Feb;1(2):302-15. doi: 10.3390/nu1020302. Epub 2009 Dec 22. Nutrients. 2009. PMID: 22253986 Free PMC article.
-
CgDM9CP-5-Integrin-MAPK Pathway Regulates the Production of CgIL-17s and Cgdefensins in the Pacific Oyster, Crassostrea gigas.J Immunol. 2023 Feb 1;210(3):245-258. doi: 10.4049/jimmunol.2200016. J Immunol. 2023. PMID: 36548464 Free PMC article.
-
Glycoprotein 2 in health and disease: lifting the veil.Genes Environ. 2021 Dec 3;43(1):53. doi: 10.1186/s41021-021-00229-8. Genes Environ. 2021. PMID: 34861888 Free PMC article. Review.
References
-
- Hoops TC, Rindler MJ. Isolation of the cDNA encoding glycoprotein-2 (GP-2), the major zymogen granule membrane protein. J BiolChem. 1991;266:4257–4263. - PubMed
-
- Dittie A, Kern H-F. The major zymogen granule membrane protein GP-2 in the rat pancreas is not involved in granule formation. Eur J Cell Biol. 1992;58:243–258. - PubMed
-
- Lowe AW, Luthen RE, Wong SME, Grendell JH. The zymogen granule protein, GP-2, is elevated in a rat model for acute pancreatitis. Gastroenterology. 1994;107:1819–1827. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

