RETRACTED: Identification of phosphorylated p38 as a novel DAPK-interacting partner during TNFalpha-induced apoptosis in colorectal tumor cells
- PMID: 19628771
- PMCID: PMC2716956
- DOI: 10.2353/ajpath.2009.080853
RETRACTED: Identification of phosphorylated p38 as a novel DAPK-interacting partner during TNFalpha-induced apoptosis in colorectal tumor cells
Retraction in
-
Retractions.Am J Pathol. 2017 Jan;187(1):225. doi: 10.1016/j.ajpath.2016.09.019. Am J Pathol. 2017. PMID: 27993239 Free PMC article. No abstract available.
Abstract
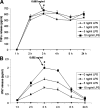
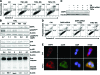
Death-associated protein kinase (DAPK) is a serine/threonine kinase that contributes to pro-apoptotic signaling on cytokine exposure. The role of DAPK in macrophage-associated tumor cell death is currently unknown. Recently, we suggested a new function for DAPK in the induction of apoptosis during the interaction between colorectal tumor cells and tumor-associated macrophages. Using a cell-culture model with conditioned supernatants of differentiated/activated macrophages (U937) and human HCT116 colorectal tumor cells, we replicated DAPK-associated tumor cell death; this model likely reflects the in vivo tumor setting. In this study, we show that tumor necrosis factor-alpha exposure under conditions of macrophage activation induced DAPK-dependent apoptosis in the colorectal tumor cell line HCT116. Simultaneously, early phosphorylation of p38 mitogen-activated protein kinase (phospho-p38) was observed. We identified the phospho-p38 mitogen-activated protein kinase as a novel interacting protein of DAPK in tumor necrosis factor-alpha-induced apoptosis. The general relevance of this interaction was verified in two colorectal cell lines without functional p53 (ie, HCT116 p53(-/-) and HT29 mutant) and in human colon cancer and ulcerative colitis tissues. Supernatants of freshly isolated human macrophages were also able to induce DAPK and phospho-p38. Our findings highlight the mechanisms that underlie DAPK regulation in tumor cell death evoked by immune cells.
Figures







References
-
- Deiss LP, Feinstein E, Berissi H, Cohen O, Kimchi A. Identification of a novel serine/threonine kinase and a novel 15-kD protein as potential mediators of the gamma interferon-induced cell death. Genes Dev. 1995;9:15–30. - PubMed
-
- Jang CW, Chen CH, Chen CC, Chen JY, Su YH, Chen RH. TGF-beta induces apoptosis through Smad-mediated expression of DAP-kinase. Nat Cell Biol. 2001;4:51–58. - PubMed
-
- Martoriati A, Doumont G, Alcalay M, Bellefroid E, Pelicci PG, Marine JC. DAPK1, encoding an activator of a p19ARF-p53 mediated apoptotic checkpoint, is a transcription target of p53. Oncogene. 2005;24:1461–1466. - PubMed
-
- Raveh T, Droguett G, Horwitz MS, DePinho RA, Kimchi A. DAP kinase activates a p19ARF/p53-mediated apoptotic checkpoint to suppress oncogenic transformation. Nat Cell Biol. 2001;3:1–7. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

