Effect of B7.1 costimulation on T-cell based immunity against TAP-negative cancer can be facilitated by TAP1 expression
- PMID: 19629186
- PMCID: PMC2711302
- DOI: 10.1371/journal.pone.0006385
Effect of B7.1 costimulation on T-cell based immunity against TAP-negative cancer can be facilitated by TAP1 expression
Abstract
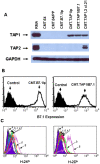
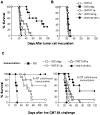
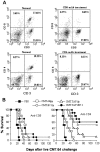
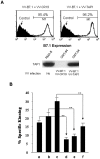
Tumors deficient in expression of the transporter associated with antigen processing (TAP) usually fail to induce T-cell-mediated immunity and are resistant to T-cell lysis. However, we have found that introduction of the B7.1 gene into TAP-negative (TAP(-)) or TAP1-transfected (TAP1(+)) murine lung carcinoma CMT.64 cells can augment the capacity of the cells to induce a protective immune response against wild-type tumor cells. Differences in the strength of the protective immune responses were observed between TAP(-) and TAP1(+) B7.1 expressing CMT.64 cells depending on the doses of gamma-irradiated cell immunization. While mice immunized with either high or low dose of B7.1-expressing TAP1(+) cells rejected TAP(-) tumors, only high dose immunization with B7.1-expressing TAP(-) cells resulted in tumor rejection. The induced protective immunity was T-cell dependent as demonstrated by dramatically reduced antitumor immunity in mice depleted of CD8 or CD4 cells. Augmentation of T-cell mediated immune response against TAP(-) tumor cells was also observed in a virally infected tumor cell system. When mice were immunized with a high dose of gamma-irradiated CMT.64 cells infected with vaccinia viruses carrying B7.1 and/or TAP1 genes, we found that the cells co-expressing B7.1 and TAP1, but not those expressing B7.1 alone, induced protective immunity against CMT.64 cells. In addition, inoculation with live tumor cells transfected with several different gene(s) revealed that only B7.1- and TAP1-coexpressing tumor cells significantly decreased tumorigenicity. These results indicate that B7.1-provoked antitumor immunity against TAP(-) cancer is facilitated by TAP1-expression, and thus both genes should be considered for cancer therapy in the future.
Conflict of interest statement
Figures

Similar articles
-
Priming of immune responses against transporter associated with antigen processing (TAP)-deficient tumours: tumour direct priming.Immunology. 2009 Nov;128(3):420-8. doi: 10.1111/j.1365-2567.2009.03127.x. Immunology. 2009. PMID: 20067541 Free PMC article.
-
Increased tumorigenicity, but unchanged immunogenicity, of transporter for antigen presentation 1-deficient tumors.Cancer Res. 2002 May 15;62(10):2856-60. Cancer Res. 2002. PMID: 12019164
-
Deficiency of transporter for antigen presentation (TAP) in tumor cells allows evasion of immune surveillance and increases tumorigenesis.J Immunol. 1999 Oct 15;163(8):4224-31. J Immunol. 1999. PMID: 10510359
-
Systemic deficits in transporter for antigen presentation (TAP)-1 or proteasome subunit LMP2 have little or no effect on tumor incidence.Int J Cancer. 2001 Feb 1;91(3):366-72. doi: 10.1002/1097-0215(200002)9999:9999<::aid-ijc1056>3.0.co;2-k. Int J Cancer. 2001. PMID: 11169961
-
B7-mediated costimulation and the immune response.Blood Rev. 1996 Jun;10(2):111-27. doi: 10.1016/s0268-960x(96)90040-5. Blood Rev. 1996. PMID: 8813343 Review.
Cited by
-
ADME gene-driven prognostic model for bladder cancer: a breakthrough in predicting survival and personalized treatment.Hereditas. 2025 Mar 19;162(1):42. doi: 10.1186/s41065-025-00409-4. Hereditas. 2025. PMID: 40108724 Free PMC article.
-
A novel category of antigens enabling CTL immunity to tumor escape variants: Cinderella antigens.Cancer Immunol Immunother. 2012 Jan;61(1):119-25. doi: 10.1007/s00262-011-1160-x. Epub 2011 Nov 25. Cancer Immunol Immunother. 2012. PMID: 22116347 Free PMC article. Review.
-
Influence of immune privilege on ocular tumor development.Ocul Immunol Inflamm. 2010 Apr;18(2):80-90. doi: 10.3109/09273941003669950. Ocul Immunol Inflamm. 2010. PMID: 20370332 Free PMC article. Review.
-
Limited density of an antigen presented by RMA-S cells requires B7-1/CD28 signaling to enhance T-cell immunity at the effector phase.PLoS One. 2014 Nov 10;9(11):e108192. doi: 10.1371/journal.pone.0108192. eCollection 2014. PLoS One. 2014. PMID: 25383875 Free PMC article.
References
-
- Melief CJ, Toes RE, Medema JP, van der Burg SH, Ossendorp F, et al. Strategies for immunotherapy of cancer. Adv Immunol. 2000;75:235–282. - PubMed
-
- Seliger B, Maeurer MJ, Ferrone S. TAP off–tumors on. Immunol Today. 1997;18:292–299. - PubMed
-
- Seliger B, Maeurer MJ, Ferrone S. Antigen-processing machinery breakdown and tumor growth. Immunol Today. 2000;21:455–464. - PubMed
-
- Garcia-Lora A, Algarra I, Garrido F. MHC class I antigens, immune surveillance, and tumor immune escape. J Cell Physiol. 2003;195:346–355. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

