Wnt/beta-catenin signaling plays an essential role in activation of odontogenic mesenchyme during early tooth development
- PMID: 19631205
- PMCID: PMC2752344
- DOI: 10.1016/j.ydbio.2009.07.015
Wnt/beta-catenin signaling plays an essential role in activation of odontogenic mesenchyme during early tooth development
Abstract
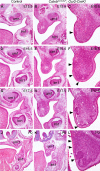
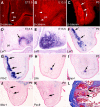
Classical tissue recombination studies demonstrated that initiation of tooth development depends on activation of odontogenic potential in the mesenchyme by signals from the presumptive dental epithelium. Although several members of the Wnt family of signaling molecules are expressed in the presumptive dental epithelium at the beginning of tooth initiation, whether Wnt signaling is directly involved in the activation of the odontogenic mesenchyme has not been characterized. In this report, we show that tissue-specific inactivation of beta-catenin, a central component of the canonical Wnt signaling pathway, in the developing tooth mesenchyme caused tooth developmental arrest at the bud stage in mice. We show that mesenchymal beta-catenin function is required for expression of Lef1 and Fgf3 in the developing tooth mesenchyme and for induction of primary enamel knot in the developing tooth epithelium. Expression of Msx1 and Pax9, two essential tooth mesenchyme transcription factors downstream of Bmp and Fgf signaling, respectively, were not altered in the absence of beta-catenin in the tooth mesenchyme. Moreover, we found that constitutive stabilization of beta-catenin in the developing palatal mesenchyme induced aberrant palatal epithelial invaginations that resembled early tooth buds both morphologically and in epithelial molecular marker expression, but without activating expression of Msx1 and Pax9 in the mesenchyme. Together, these results indicate that activation of the mesenchymal odontogenic program during early tooth development requires concerted actions of Bmp, Fgf and Wnt signaling from the presumptive dental epithelium to the mesenchyme.
Figures
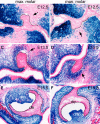
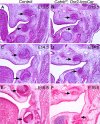

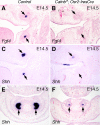

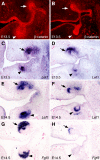

References
-
- Andl T, Reddy ST, Gaddapara T, Millar SE. WNT signals are required for the initiation of hair follicle development. Dev. Cell. 2002;2:643–653. - PubMed
-
- Andl T, Ahn K, Kairo A, Chu EY, Wine-Lee L, Reddy ST, Croft NJ, Cebra-Thomas JA, Metzger D, Chambon P, Lyons KM, Mishina Y, Seykora JT, Crenshaw EB, III, Millar SE. Epithelial Bmpr1a regulates differentiation and proliferation in postnatal hair follicles and is essential for tooth development. Development. 2004;131:2257–2268. - PubMed
-
- Barolo S. Transgenic Wnt/TCF pathway reporters: all you need is Lef? Oncogene. 2006;25:7505–7511. - PubMed
-
- Bei M, Maas R. FGFs and BMP4 induce both Msx1-independent and Msx1-dependent signaling pathways in early tooth development. Development. 1998;125:4325–4333. - PubMed
-
- Bei M, Kratochwil K, Maas RL. BMP4 rescues a non-cell-autonomous function of Msx1 in tooth development. Development. 2000;127:4711–4718. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

