The Ca2+ affinity of synaptotagmin 1 is markedly increased by a specific interaction of its C2B domain with phosphatidylinositol 4,5-bisphosphate
- PMID: 19632983
- PMCID: PMC2757977
- DOI: 10.1074/jbc.M109.042499
The Ca2+ affinity of synaptotagmin 1 is markedly increased by a specific interaction of its C2B domain with phosphatidylinositol 4,5-bisphosphate
Abstract
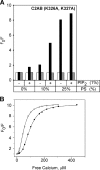
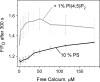
The synaptic vesicle protein synaptotagmin 1 is thought to convey the calcium signal onto the core secretory machinery. Its cytosolic portion mainly consists of two C2 domains, which upon calcium binding are enabled to bind to acidic lipid bilayers. Despite major advances in recent years, it is still debated how synaptotagmin controls the process of neurotransmitter release. In particular, there is disagreement with respect to its calcium binding properties and lipid preferences. To investigate how the presence of membranes influences the calcium affinity of synaptotagmin, we have now measured these properties under equilibrium conditions using isothermal titration calorimetry and fluorescence resonance energy transfer. Our data demonstrate that the acidic phospholipid phosphatidylinositol 4,5-bisphosphate (PI(4,5)P2), but not phosphatidylserine, markedly increases the calcium sensitivity of synaptotagmin. PI(4,5)P2 binding is confined to the C2B domain but is not affected significantly by mutations of a lysine-rich patch. Together, our findings lend support to the view that synaptotagmin functions by binding in a trans configuration whereby the C2A domain binds to the synaptic vesicle and the C2B binds to the PI(4,5)P2-enriched plasma membrane.
Figures

References
-
- Sudhof T. C. (2004) Annu. Rev. Neurosci. 27, 509–547 - PubMed
-
- Jahn R., Scheller R. H. (2006) Nat. Rev. Mol. Cell Biol. 7, 631–643 - PubMed
-
- Rizo J., Chen X., Araç D. (2006) Trends Cell Biol. 16, 339–350 - PubMed
-
- Wojcik S. M., Brose N. (2007) Neuron 55, 11–24 - PubMed
-
- Chapman E. R. (2008) Annu. Rev. Biochem. 77, 615–641 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

