Systemic dysregulation of CEACAM1 in melanoma patients
- PMID: 19633846
- PMCID: PMC11030926
- DOI: 10.1007/s00262-009-0740-5
Systemic dysregulation of CEACAM1 in melanoma patients
Abstract
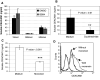
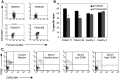
It was previously shown that CEACAM1 on melanoma cells strongly predicts poor outcome. Here, we show a statistically significant increase of serum CEACAM1 in 64 active melanoma patients, as compared to 48 patients with no evidence of disease and 37 healthy donors. Among active patients, higher serum CEACAM1 correlated with LDH values and with decreased survival. Multivariate analysis with neutralization of LDH showed that increased serum CEACAM1 carries a hazard ratio of 2.40. In vitro, soluble CEACAM1 was derived from CEACAM1(+), but neither from CEACAM1(-) melanoma cells nor from CEACAM1(+) lymphocytes, and directly correlated with the number of CEACAM1(+) melanoma cells. Production of soluble CEACAM1 depended on intact de novo protein synthesis and secretion machineries, but not on metalloproteinase function. An unusually high percentage of CEACAM1(+) circulating NK and T lymphocytes was demonstrated in melanoma patients. CEACAM1 inhibited killing activity in functional assays. CEACAM1 expression could not be induced on lymphocytes by serum from patients with high CEACAM1 expression. Further, expression of other NK receptors was impaired, which collectively indicate on a general abnormality. In conclusion, the systemic dysregulation of CEACAM1 in melanoma patients further denotes the role of CEACAM1 in melanoma and may provide a basis for new tumor monitoring and prognostic platforms.
Figures






Similar articles
-
The human antibody fragment DIATHIS1 specific for CEACAM1 enhances natural killer cell cytotoxicity against melanoma cell lines in vitro.J Immunother. 2015 Nov-Dec;38(9):357-70. doi: 10.1097/CJI.0000000000000100. J Immunother. 2015. PMID: 26448580 Free PMC article.
-
CEACAM1-3S Drives Melanoma Cells into NK Cell-Mediated Cytolysis and Enhances Patient Survival.Cancer Res. 2015 May 1;75(9):1897-907. doi: 10.1158/0008-5472.CAN-14-1752. Epub 2015 Mar 5. Cancer Res. 2015. PMID: 25744717
-
CEACAM1 in malignant melanoma: a diagnostic and therapeutic target.Curr Top Med Chem. 2012;12(1):3-10. doi: 10.2174/156802612798919259. Curr Top Med Chem. 2012. PMID: 22196267 Review.
-
Serum CEACAM1 Correlates with Disease Progression and Survival in Malignant Melanoma Patients.Clin Dev Immunol. 2012;2012:290536. doi: 10.1155/2012/290536. Epub 2012 Jan 16. Clin Dev Immunol. 2012. PMID: 22291846 Free PMC article.
-
CEACAM1 and the regulation of mucosal inflammation.Mucosal Immunol. 2008 Nov;1 Suppl 1(0 1):S39-42. doi: 10.1038/mi.2008.50. Mucosal Immunol. 2008. PMID: 19079227 Free PMC article. Review.
Cited by
-
CEACAM1: Expression and Role in Melanocyte Transformation.Dis Markers. 2016;2016:9406319. doi: 10.1155/2016/9406319. Epub 2016 Aug 24. Dis Markers. 2016. PMID: 27642217 Free PMC article. Review.
-
Glycosylation Alters Dimerization Properties of a Cell-surface Signaling Protein, Carcinoembryonic Antigen-related Cell Adhesion Molecule 1 (CEACAM1).J Biol Chem. 2016 Sep 16;291(38):20085-95. doi: 10.1074/jbc.M116.740050. Epub 2016 Jul 28. J Biol Chem. 2016. PMID: 27471271 Free PMC article.
-
CEACAM1 and MICA as novel serum biomarkers in patients with acute and recurrent pericarditis.Oncotarget. 2016 Apr 5;7(14):17885-95. doi: 10.18632/oncotarget.7530. Oncotarget. 2016. PMID: 26909604 Free PMC article.
-
CEACAM1-4L Promotes Anchorage-Independent Growth in Melanoma.Front Oncol. 2015 Oct 19;5:234. doi: 10.3389/fonc.2015.00234. eCollection 2015. Front Oncol. 2015. PMID: 26539411 Free PMC article.
-
Development of allogeneic NK cell adoptive transfer therapy in metastatic melanoma patients: in vitro preclinical optimization studies.PLoS One. 2013;8(3):e57922. doi: 10.1371/journal.pone.0057922. Epub 2013 Mar 4. PLoS One. 2013. PMID: 23483943 Free PMC article.
References
-
- National Cancer Institute. Surveillance epidemiology and end results. http://seer.cancer.gov/csr/1975_2004/results_merged/sect_16_melanoma.pdf. Last accessed at 28 January 2009
-
- National Cancer Institute. Surveillance epidemiology and end results. http://seer.cancer.gov/publications/aya/5_melanoma.pdf. Last accessed at 28 January 2009
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

