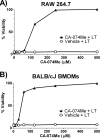
CA-074Me protection against anthrax lethal toxin
- PMID: 19635822
- PMCID: PMC2747948
- DOI: 10.1128/IAI.00730-09
CA-074Me protection against anthrax lethal toxin
Abstract
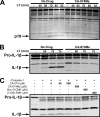
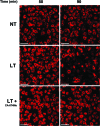
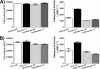
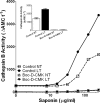
Anthrax lethal toxin (LT) activates the NLRP1b (NALP1b) inflammasome and caspase-1 in macrophages from certain inbred mouse strains, but the mechanism by which this occurs is poorly understood. We report here that similar to several NLRP3 (NALP3, cryopyrin)-activating stimuli, LT activation of the NLRP1b inflammasome involves lysosomal membrane permeabilization (LMP) and subsequent cytoplasmic cathepsin B activity. CA-074Me, a potent cathepsin B inhibitor, protects LT-sensitive macrophages from cell death and prevents the activation of caspase-1. RNA interference knockdown of cathepsin B expression, however, cannot prevent LT-mediated cell death, suggesting that CA-074Me may also act on other cellular proteases released during LMP. CA-074Me appears to function downstream of LT translocation to the cytosol (as assessed by mitogen-activated protein kinase kinase cleavage), K(+) effluxes, and proteasome activity. The initial increase in cytoplasmic activity of cathepsin B occurs at the same time or shortly before caspase-1 activation but precedes a larger-scale lysosomal destabilization correlated closely with cytolysis. We present results suggesting that LMP may be involved in the activation of the NLRP1b inflammasome.
Figures




References
-
- Alileche, A., R. C. Squires, S. M. Muehlbauer, M. P. Lisanti, and J. Brojatsch. 2006. Mitochondrial impairment is a critical event in anthrax lethal toxin-induced cytolysis of murine macrophages. Cell Cycle 5:100-106. - PubMed
-
- Benchoua, A., J. Braudeau, A. Reis, C. Couriaud, and B. Onteniente. 2004. Activation of proinflammatory caspases by cathepsin B in focal cerebral ischemia. J. Cereb. Blood Flow Metab. 24:1272-1279. - PubMed
-
- Bidere, N., H. K. Lorenzo, S. Carmona, M. Laforge, F. Harper, C. Dumont, and A. Senik. 2003. Cathepsin D triggers Bax activation, resulting in selective apoptosis-inducing factor (AIF) relocation in T lymphocytes entering the early commitment phase to apoptosis. J. Biol. Chem. 278:31401-31411. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

