Induction of antigen-specific tolerance by oral administration of Lactococcus lactis delivered immunodominant DQ8-restricted gliadin peptide in sensitized nonobese diabetic Abo Dq8 transgenic mice
- PMID: 19635921
- PMCID: PMC3480315
- DOI: 10.4049/jimmunol.0802891
Induction of antigen-specific tolerance by oral administration of Lactococcus lactis delivered immunodominant DQ8-restricted gliadin peptide in sensitized nonobese diabetic Abo Dq8 transgenic mice
Abstract
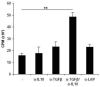
Active delivery of recombinant autoantigens or allergens at the intestinal mucosa by genetically modified Lactococcus lactis (LL) provides a novel therapeutic approach for the induction of tolerance. Celiac disease is associated with either HLA-DQ2- or HLA-DQ8-restricted responses to specific antigenic epitopes of gliadin, and may be treated by induction of Ag-specific tolerance. We investigated whether oral administration of LL-delivered DQ8-specific gliadin epitope induces Ag-specific tolerance. LL was engineered to secrete a deamidated DQ8 gliadin epitope (LL-eDQ8d) and the induction of Ag-specific tolerance was studied in NOD AB degrees DQ8 transgenic mice. Tolerance was assessed by delayed-type hypersensitivity reaction, cytokine measurements, eDQ8d-specific proliferation, and regulatory T cell analysis. Oral administration of LL-eDQ8d induced suppression of local and systemic DQ8-restricted T cell responses in NOD AB degrees DQ8 transgenic mice. Treatment resulted in an Ag-specific decrease of the proliferative capacity of inguinal lymph node (ILN) cells and lamina propria cells. Production of IL-10 and TGF-beta and a significant induction of Foxp3(+) regulatory T cells were associated with the eDQ8d-specific suppression induced by LL-eDQ8d. These data provide support for the development of effective therapeutic approaches for gluten-sensitive disorders using orally administered Ag-secreting LL. Such treatments may be effective even in the setting of established hypersensitivity.
Figures
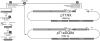
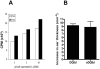
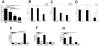
References
-
- Stepniak D, Koning F. Celiac disease--sandwiched between innate and adaptive immunity. Hum Immunol. 2006;67:460. - PubMed
-
- Sollid LM, Khosla C. Future therapeutic options for celiac disease. Nat Clin Pract Gastroenterol Hepatol. 2005;2:140. - PubMed
-
- Jabri B, Sollid LM. Mechanisms of disease: immunopathogenesis of celiac disease. Nat Clin Pract Gastroenterol Hepatol. 2006;3:516. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

