Surface feature-guided mapping of cerebral metabolic changes in cognitively normal and mildly impaired elderly
- PMID: 19636640
- PMCID: PMC2844536
- DOI: 10.1007/s11307-009-0247-7
Surface feature-guided mapping of cerebral metabolic changes in cognitively normal and mildly impaired elderly
Abstract
Purpose: The aim of this study was to investigate the longitudinal positron emission tomography (PET) metabolic changes in the elderly.
Procedures: Nineteen nondemented subjects (mean Mini-Mental Status Examination 29.4 +/- 0.7 SD) underwent two detailed neuropsychological evaluations and resting 2-deoxy-2-[F-18]fluoro-D: -glucose (FDG)-PET scan (interval 21.7 +/- 3.7 months), baseline structural 3T magnetic resonance (MR) imaging, and apolipoprotein E4 genotyping. Cortical PET metabolic changes were analyzed in 3-D using the cortical pattern matching technique.
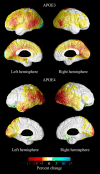
Results: Baseline vs. follow-up whole-group comparison revealed significant metabolic decline bilaterally in the posterior temporal, parietal, and occipital lobes and the left lateral frontal cortex. The declining group demonstrated 10-15% decline in bilateral posterior cingulate/precuneus, posterior temporal, parietal, and occipital cortices. The cognitively stable group showed 2.5-5% similarly distributed decline. ApoE4-positive individuals underwent 5-15% metabolic decline in the posterior association cortices.
Conclusions: Using 3-D surface-based MR-guided FDG-PET mapping, significant metabolic changes were seen in five posterior and the left lateral frontal regions. The changes were more pronounced for the declining relative to the cognitively stable group.
Figures


References
-
- Silverman DHS, Thompson PM (2006) Structural and functional neuroimaging: focussing on mild cognitive impairment. Applied Neurology 10-24
-
- Drzezga A, Grimmer T, Riemenschneider M, et al. Prediction of individual clinical outcome in MCI by means of genetic assessment and (18)F-FDG-PET. J Nucl Med. 2005;46:1625–1632. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- P01-AG024831/AG/NIA NIH HHS/United States
- P50 AG16570/AG/NIA NIH HHS/United States
- P30 AG010123/AG/NIA NIH HHS/United States
- EB01651/EB/NIBIB NIH HHS/United States
- R01 AG013308/AG/NIA NIH HHS/United States
- R01 MH052453/MH/NIMH NIH HHS/United States
- K23 AG026803/AG/NIA NIH HHS/United States
- P41 RR013642/RR/NCRR NIH HHS/United States
- U54 RR021813/RR/NCRR NIH HHS/United States
- R01 MH058156/MH/NIMH NIH HHS/United States
- LM05639/LM/NLM NIH HHS/United States
- M01-RR00865/RR/NCRR NIH HHS/United States
- R01 MH071940/MH/NIMH NIH HHS/United States
- AG 13308/AG/NIA NIH HHS/United States
- MH/AG58156/AG/NIA NIH HHS/United States
- R21 RR019771/RR/NCRR NIH HHS/United States
- R01 LM005639/LM/NLM NIH HHS/United States
- MH52453/MH/NIMH NIH HHS/United States
- M01 RR000865/RR/NCRR NIH HHS/United States
- RR019771/RR/NCRR NIH HHS/United States
- P50 AG016570/AG/NIA NIH HHS/United States

