Surface binding inhibitors of the SCF-KIT protein-protein interaction
- PMID: 19637142
- PMCID: PMC2805069
- DOI: 10.1002/cbic.200900079
Surface binding inhibitors of the SCF-KIT protein-protein interaction
Figures
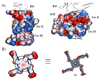
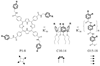
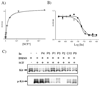
 ) and P3 (●); KI(app) = 31 and 33 n
) and P3 (●); KI(app) = 31 and 33 nReferences
-
- Ashman LK. Int. J. Biochem. Cell Biol. 1999;31:1037–1051. - PubMed
-
- Lemmon MA, Pinchasi D, Zhou M, Lax I, Schlessinger J. J. Biol. Chem. 1997;272:6311–6317. - PubMed
-
- Hirota S, Isozaki K, Moriyama Y, Hashimoto K, Nishida T, Ishiguro S, Kawano K, Hanada M, Kurata A, Takeda M, Tunio GM, Matsuzawa Y, Kanakura Y, Shinomura Y, Kitamura Y. Science. 1998;279:577–580. - PubMed
-
- Nishida T, Hirota S, Taniguchi M, Hashimoto K, Isozaki K, Nakamura H, Kanakura Y, Tanaka T, Takabayashi A, Matsuda H, Kitamura Y. Nat. Genet. 1998;19:323–324. - PubMed
-
- Zheng R, Klang K, Gorin N, Small D. Leuk. Res. 2004;28:121–126. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

