Squaraine rotaxanes with boat conformation macrocycles
- PMID: 19639940
- PMCID: PMC2847772
- DOI: 10.1021/jo901298n
Squaraine rotaxanes with boat conformation macrocycles
Abstract
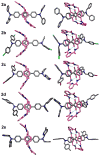
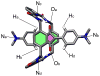
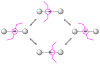
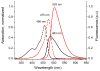
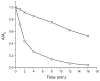
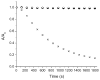
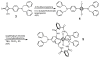
Mechanical encapsulation of fluorescent, deep-red bis(anilino)squaraine dyes inside Leigh-type tetralactam macrocycles produces interlocked squaraine rotaxanes. The surrounding macrocycles are flexible and undergo rapid exchange of chair and boat conformations in solution. A series of X-ray crystal structures show how the rotaxane co-conformational exchange process involves simultaneous lateral oscillation of the macrocycle about the center of the encapsulated squaraine thread. Rotaxane macrocycles with 1,4-phenylene sidewalls and 2,6-pyridine dicarboxamide bridging units are more likely to adopt boat conformations in the solid state than analogous squaraine rotaxane systems with isophthalamide-containing macrocycles. A truncated squaraine dye, with a secondary amine attached directly to the central C(4)O(2) core, is less electrophilic than the extended bis(anilino)squaraine analogue, but it is still susceptible to chemical and photochemical bleaching. Its stability is greatly enhanced when it is encapsulated as an interlocked squaraine rotaxane. An X-ray crystal structure of this truncated squaraine rotaxane shows the macrocycle in a boat conformation, and NMR studies indicate that the boat is maintained in solution. Encapsulation as a rotaxane increases the dye's brightness by a factor of 6. The encapsulation process appears to constrain the dye and reduce deformation of the chromophore from planarity. This study shows how mechanical encapsulation as a rotaxane can be used as a rational design parameter to fine-tune the chemical and photochemical properties of squaraine dyes.
Figures
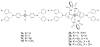

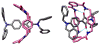
Similar articles
-
Synthesis and photophysical investigation of squaraine rotaxanes by "clicked capping".Org Lett. 2008 Aug 7;10(15):3343-6. doi: 10.1021/ol801189a. Epub 2008 Jun 27. Org Lett. 2008. PMID: 18582079 Free PMC article.
-
Squaraine-derived rotaxanes: highly stable, fluorescent near-IR dyes.Chemistry. 2006 Jun 2;12(17):4684-90. doi: 10.1002/chem.200501541. Chemistry. 2006. PMID: 16575935
-
Squaraine-derived rotaxanes: sterically protected fluorescent near-IR dyes.J Am Chem Soc. 2005 Mar 16;127(10):3288-9. doi: 10.1021/ja042404n. J Am Chem Soc. 2005. PMID: 15755140
-
Squaraine Dyes: Molecular Design for Different Applications and Remaining Challenges.Bioconjug Chem. 2020 Feb 19;31(2):194-213. doi: 10.1021/acs.bioconjchem.9b00482. Epub 2019 Aug 12. Bioconjug Chem. 2020. PMID: 31365819 Free PMC article. Review.
-
Biomimetic Synchronized Motion of Two Interacting Macrocycles in [3]Rotaxane-Based Molecular Shuttles.Angew Chem Int Ed Engl. 2019 Oct 14;58(42):15136-15141. doi: 10.1002/anie.201910318. Epub 2019 Sep 9. Angew Chem Int Ed Engl. 2019. PMID: 31436864 Review.
Cited by
-
Molecular recognition using tetralactam macrocycles with parallel aromatic sidewalls.Beilstein J Org Chem. 2019 May 9;15:1086-1095. doi: 10.3762/bjoc.15.105. eCollection 2019. Beilstein J Org Chem. 2019. PMID: 31164945 Free PMC article. Review.
-
2,3-Dibutoxynaphthalene-based tetralactam macrocycles for recognizing precious metal chloride complexes.Beilstein J Org Chem. 2019 Jul 2;15:1460-1467. doi: 10.3762/bjoc.15.146. eCollection 2019. Beilstein J Org Chem. 2019. PMID: 31354862 Free PMC article.
-
Large-Amplitude Conformational Changes in Self-Assembled Multi-Stranded Aromatic Sheets.Angew Chem Int Ed Engl. 2021 Feb 1;60(5):2574-2577. doi: 10.1002/anie.202014670. Epub 2020 Nov 30. Angew Chem Int Ed Engl. 2021. PMID: 33156974 Free PMC article.
-
Rotaxane nanomachines in future molecular electronics.Nanoscale Adv. 2022 Jun 24;4(17):3418-3461. doi: 10.1039/d2na00057a. eCollection 2022 Aug 23. Nanoscale Adv. 2022. PMID: 36134345 Free PMC article. Review.
-
New Class of Hydroxy-Substituted Squaraine Rotaxane.Aust J Chem. 2010;63(5):792-796. doi: 10.1071/CH09671. Aust J Chem. 2010. PMID: 20634969 Free PMC article.
References
-
- Rescifina A, Zagni C, Iannazzo D, Merino P. Curr Org Chem. 2009;13:448–481.
- Kay RE, Leigh DA. Pure Appl Chem. 2008;80:17–29.
- Griffiths KE, Stoddart JF. Pure Appl Chem. 2008;80:485–506.
- Kay ER, Leigh DA, Zerbetto F. Angew Chem Int Ed. 2007;46:72–191. - PubMed
- Loeb SJ. Chem Soc Rev. 2007;36:226–235. - PubMed
- Tian H, Wang QC. Chem Soc Rev. 2006;35:361–374. - PubMed
- Schalley CA, Weilandt T, Bruggemann J, Vögtle F. Top Curr Chem. 2005;248:141–200.
-
- Betancourt JE, Martin-Hidalgo M, Gubala V, Rivera JM. J Am Chem Soc. 2009;131:3186–3188. - PMC - PubMed
- Zhu SS, Nieger M, Daniels J, Felder T, Kossev I, Schmidt T, Sokolowski M, Vogtle F, Schalley CA. Chem Eur J. 2009;15:5040–5046. - PubMed
- Pons M, Millet O. Prog Nucl Magn Reson Spectrosc. 2001;38:267–324.
-
- Leigh DA, Murphy A, Smart JP, Slawin AMZ. Angew Chem Int Ed. 1997;36:728–732.
- Gatti FG, Leigh DA, Nepogodiev SA, Slawin AMZ, Teat SJ, Wong JKY. J Am Chem Soc. 2001;123:5983–5989. - PubMed
- Lane AS, Leigh DA, Murphy A. J Am Chem Soc. 1997;119:11092–11093.
- Clegg W, Gimenez-Saiz C, Leigh DA, Murphy A, Slawin AMZ, Teat SJ. J Am Chem Soc. 1999;121:4124–4129.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

