Pyridoxal 5'-phosphate: electrophilic catalyst extraordinaire
- PMID: 19640775
- PMCID: PMC2749917
- DOI: 10.1016/j.cbpa.2009.06.023
Pyridoxal 5'-phosphate: electrophilic catalyst extraordinaire
Abstract
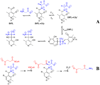
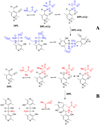
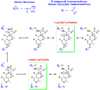
Studies of nonenzymatic electrophilic catalysis of carbon deprotonation of glycine show that pyridoxal 5'-phosphate (PLP) strongly enhances the carbon acidity of alpha-amino acids, but that this is not the overriding mechanistic imperative for cofactor catalysis. Although the fully protonated PLP-glycine iminium ion adduct exhibits an extraordinary low alpha-imino carbon acidity (pK(a)=6), the more weakly acidic zwitterionic iminium ion adduct (pK(a)=17) is selected for use in enzymatic reactions. The similar alpha-imino carbon acidities of the iminium ion adducts of glycine with 5'-deoxypyridoxal and with phenylglyoxylate show that the cofactor pyridine nitrogen plays a relatively minor role in carbanion stabilization. The 5'-phosphodianion group of PLP likely plays an important role in catalysis by providing up to 12 kcal/mol of binding energy that may be utilized for transition state stabilization.
Figures

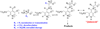

Similar articles
-
The PLP cofactor: lessons from studies on model reactions.Biochim Biophys Acta. 2011 Nov;1814(11):1419-25. doi: 10.1016/j.bbapap.2010.12.007. Epub 2010 Dec 20. Biochim Biophys Acta. 2011. PMID: 21182991 Free PMC article. Review.
-
Substituent effects on electrophilic catalysis by the carbonyl group: anatomy of the rate acceleration for PLP-catalyzed deprotonation of glycine.J Am Chem Soc. 2011 Mar 9;133(9):3173-83. doi: 10.1021/ja110795m. Epub 2011 Feb 16. J Am Chem Soc. 2011. PMID: 21323335 Free PMC article.
-
Covalent catalysis by pyridoxal: evaluation of the effect of the cofactor on the carbon acidity of glycine.J Am Chem Soc. 2007 Mar 14;129(10):3013-21. doi: 10.1021/ja0679228. Epub 2007 Feb 14. J Am Chem Soc. 2007. PMID: 17298067 Free PMC article.
-
Glycine enolates: the effect of formation of iminium ions to simple ketones on alpha-amino carbon acidity and a comparison with pyridoxal iminium ions.J Am Chem Soc. 2008 Feb 13;130(6):2041-50. doi: 10.1021/ja078006c. Epub 2008 Jan 17. J Am Chem Soc. 2008. PMID: 18198876 Free PMC article.
-
Molecular dynamics simulations of the intramolecular proton transfer and carbanion stabilization in the pyridoxal 5'-phosphate dependent enzymes L-dopa decarboxylase and alanine racemase.Biochim Biophys Acta. 2011 Nov;1814(11):1438-46. doi: 10.1016/j.bbapap.2011.05.002. Epub 2011 May 10. Biochim Biophys Acta. 2011. PMID: 21600315 Free PMC article. Review.
Cited by
-
NMR Crystallography of a Carbanionic Intermediate in Tryptophan Synthase: Chemical Structure, Tautomerization, and Reaction Specificity.J Am Chem Soc. 2016 Nov 23;138(46):15214-15226. doi: 10.1021/jacs.6b08937. Epub 2016 Nov 11. J Am Chem Soc. 2016. PMID: 27779384 Free PMC article.
-
Structures and kinetics of Thermotoga maritima MetY reveal new insights into the predominant sulfurylation enzyme of bacterial methionine biosynthesis.J Biol Chem. 2021 Jan-Jun;296:100797. doi: 10.1016/j.jbc.2021.100797. Epub 2021 May 18. J Biol Chem. 2021. PMID: 34019879 Free PMC article.
-
Aspartate aminotransferase: an old dog teaches new tricks.Arch Biochem Biophys. 2014 Feb 15;544:119-27. doi: 10.1016/j.abb.2013.10.002. Epub 2013 Oct 9. Arch Biochem Biophys. 2014. PMID: 24121043 Free PMC article. Review.
-
Controlling reaction specificity in pyridoxal phosphate enzymes.Biochim Biophys Acta. 2011 Nov;1814(11):1407-18. doi: 10.1016/j.bbapap.2011.05.019. Epub 2011 Jun 6. Biochim Biophys Acta. 2011. PMID: 21664990 Free PMC article. Review.
-
Theoretical and Mechanistic Validation of Global Kinetic Parameters of the Inactivation of GABA Aminotransferase by OV329 and CPP-115.ACS Chem Biol. 2021 Apr 16;16(4):615-630. doi: 10.1021/acschembio.0c00784. Epub 2021 Mar 18. ACS Chem Biol. 2021. PMID: 33735567 Free PMC article.
References
-
- Snell EE, Braunstein AE, Severin ES, Torchinsky YM, editors. Pyridoxal Catalysis Enzymes and Model Systems. New York: Interscience; 1968.
-
-
Barends TRM, Dunn MF, Schlichting I. Tryptophan synthase, an allosteric molecular factory. Curr.Opin.Chem.Biol. 2008;12:593–600. •• A summary of fascinating experiments that characterize the mechanism for allosteric regulation of substrate channelling at tryptophan synthase and of the reactions catalyzed by the α and β subunits.
-
-
- He X, Liu H-W. Mechanisms of enzymatic C–O bond cleavages in deoxyhexose biosynthesis. Curr.Opin.Chem.Biol. 2002;6:590–597. - PubMed
-
- Schirch V, Szebenyi DME. Serine hydroxymethyltransferase revisited. Curr.Opin.Chem.Biol. 2005;9:482–487. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

