The expression of N-terminal deletion DNA pilot proteins inhibits the early stages of phiX174 replication
- PMID: 19640994
- PMCID: PMC2748053
- DOI: 10.1128/JVI.01077-09
The expression of N-terminal deletion DNA pilot proteins inhibits the early stages of phiX174 replication
Abstract
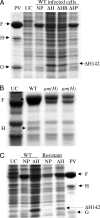
The phiX174 DNA pilot protein H contains four predicted C-terminal coiled-coil domains. The region of the gene encoding these structures was cloned, expressed in vivo, and found to strongly inhibit wild-type replication. DNA and protein synthesis was investigated in the absence of de novo H protein synthesis and in wild-type-infected cells expressing the inhibitory proteins (DeltaH). The expression of the DeltaH proteins interfered with early stages of DNA replication, which did not require de novo H protein synthesis, suggesting that the inhibitory proteins interfere with the wild-type H protein that enters the cell with the penetrating DNA. As transcription and protein synthesis are dependent on DNA replication in positive single-stranded DNA life cycles, viral protein synthesis was also reduced. However, unlike DNA synthesis, efficient viral protein synthesis required de novo H protein synthesis, a novel function for this protein. A single amino acid change in the C terminus of protein H was both necessary and sufficient to confer resistance to the inhibitory DeltaH proteins, restoring both DNA and protein synthesis to wild-type levels. DeltaH proteins derived from the resistant mutant did not inhibit wild-type or resistant mutant replication. The inhibitory effects of the DeltaH proteins were lessened by the coexpression of the internal scaffolding protein, which may suppress H-H protein interactions. While coexpression relieved the block in DNA biosynthesis, viral protein synthesis remained suppressed. These data indicate that protein H's role in DNA replication and stimulating viral protein synthesis can be uncoupled.
Figures

Similar articles
-
Herpes simplex virus type 1 ICP0 plays a critical role in the de novo synthesis of infectious virus following transfection of viral DNA.J Virol. 1989 Nov;63(11):4579-89. doi: 10.1128/JVI.63.11.4579-4589.1989. J Virol. 1989. PMID: 2552142 Free PMC article.
-
Studies on the role of the phi X174 gene A protein in phi X174 viral strand synthesis. III. Replication of DNA containing two viral replication origins.J Biol Chem. 1988 Nov 5;263(31):16443-51. J Biol Chem. 1988. PMID: 2972713
-
Effect of phi X C protein on leading strand DNA synthesis in the phi X174 replication pathway.J Biol Chem. 1988 Nov 5;263(31):16452-60. J Biol Chem. 1988. PMID: 2972714
-
Mechanism of replication of bacteriophage phi X174. XXII. Site-specific mutagenesis of the A* gene reveals that A* protein is not essential for phi X174 DNA replication.J Mol Biol. 1987 Sep 5;197(1):47-54. doi: 10.1016/0022-2836(87)90608-5. J Mol Biol. 1987. PMID: 2960819
-
DNA sequences which support activities of the bacteriophage phi X174 gene A protein.J Biol Chem. 1983 Jul 10;258(13):8402-12. J Biol Chem. 1983. PMID: 6223031
Cited by
-
Effects of an early conformational switch defect during ϕX174 morphogenesis are belatedly manifested late in the assembly pathway.J Virol. 2013 Mar;87(5):2518-25. doi: 10.1128/JVI.02839-12. Epub 2012 Dec 19. J Virol. 2013. PMID: 23255785 Free PMC article.
-
Defensive hypervariable regions confer superinfection exclusion in microviruses.Proc Natl Acad Sci U S A. 2021 Jul 13;118(28):e2102786118. doi: 10.1073/pnas.2102786118. Proc Natl Acad Sci U S A. 2021. PMID: 34244443 Free PMC article.
-
The impact of spatial structure on viral genomic diversity generated during adaptation to thermal stress.PLoS One. 2014 Feb 12;9(2):e88702. doi: 10.1371/journal.pone.0088702. eCollection 2014. PLoS One. 2014. PMID: 24533140 Free PMC article.
-
Structure-Function Analysis of the ϕX174 DNA-Piloting Protein Using Length-Altering Mutations.J Virol. 2016 Aug 12;90(17):7956-66. doi: 10.1128/JVI.00914-16. Print 2016 Sep 1. J Virol. 2016. PMID: 27356899 Free PMC article.
-
Mutagenic Analysis of a DNA Translocating Tube's Interior Surface.Viruses. 2020 Jun 22;12(6):670. doi: 10.3390/v12060670. Viruses. 2020. PMID: 32580341 Free PMC article.
References
-
- Alfadhli, A., E. Steel, L. Finlay, H. P. Bachinger, and E. Barklis. 2002. Hantavirus nucleocapsid protein coiled-coil domains. J. Biol. Chem. 277:27103-27108. - PubMed
-
- Azuma, J., J. Morita, and T. Komano. 1980. Process of attachment of φX174 parental DNA to the host cell membrane. J. Biochem. 88:525-532. - PubMed
-
- Bayer, M. E., and T. W. Starkey. 1972. The adsorption of bacteriophage φX174 and its interaction with Escherichia coli: a kinetic and morphological study. Virology 49:236-256. - PubMed
-
- Burch, A. D., J. Ta, and B. A. Fane. 1999. Cross-functional analysis of the Microviridae internal scaffolding protein. J. Mol. Biol. 286:95-104. - PubMed
-
- Chen, M., A. Uchiyama, and B. A. Fane. 2007. Eliminating the requirement of an essential gene product in an already very small virus: scaffolding protein B-free φX174, B-free. J. Mol. Biol. 373:308-314. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

