Impairment of bidirectional synaptic plasticity in the striatum of a mouse model of DYT1 dystonia: role of endogenous acetylcholine
- PMID: 19641103
- PMCID: PMC2766181
- DOI: 10.1093/brain/awp194
Impairment of bidirectional synaptic plasticity in the striatum of a mouse model of DYT1 dystonia: role of endogenous acetylcholine
Abstract
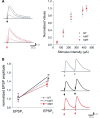
DYT1 dystonia is a severe form of inherited dystonia, characterized by involuntary twisting movements and abnormal postures. It is linked to a deletion in the dyt1 gene, resulting in a mutated form of the protein torsinA. The penetrance for dystonia is incomplete, but both clinically affected and non-manifesting carriers of the DYT1 mutation exhibit impaired motor learning and evidence of altered motor plasticity. Here, we characterized striatal glutamatergic synaptic plasticity in transgenic mice expressing either the normal human torsinA or its mutant form, in comparison to non-transgenic (NT) control mice. Medium spiny neurons recorded from both NT and normal human torsinA mice exhibited normal long-term depression (LTD), whereas in mutant human torsinA littermates LTD could not be elicited. In addition, although long-term potentiation (LTP) could be induced in all the mice, it was greater in magnitude in mutant human torsinA mice. Low-frequency stimulation (LFS) can revert potentiated synapses to resting levels, a phenomenon termed synaptic depotentiation. LFS induced synaptic depotentiation (SD) both in NT and normal human torsinA mice, but not in mutant human torsinA mice. Since anti-cholinergic drugs are an effective medical therapeutic option for the treatment of human dystonia, we reasoned that an excess in endogenous acetylcholine could underlie the synaptic plasticity impairment. Indeed, both LTD and SD were rescued in mutant human torsinA mice either by lowering endogenous acetylcholine levels or by antagonizing muscarinic M1 receptors. The presence of an enhanced acetylcholine tone was confirmed by the observation that acetylcholinesterase activity was significantly increased in the striatum of mutant human torsinA mice, as compared with both normal human torsinA and NT littermates. Moreover, we found similar alterations of synaptic plasticity in muscarinic M2/M4 receptor knockout mice, in which an increased striatal acetylcholine level has been documented. The loss of LTD and SD on one hand, and the increase in LTP on the other, demonstrate that a 'loss of inhibition' characterizes the impairment of synaptic plasticity in this model of DYT1 dystonia. More importantly, our results indicate that an unbalanced cholinergic transmission plays a pivotal role in these alterations, providing a clue to understand the ability of anticholinergic agents to restore motor deficits in dystonia.
Figures

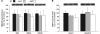




References
-
- Albin RL, Young AB, Penney JB. The functional anatomy of basal ganglia disorders. Trends Neurosci. 1989;12:366–75. - PubMed
-
- Anderson WW, Collingridge GL. Capabilities of the WinLTP data acquisition program extending beyond basic LTP experimental functions. J Neurosci Methods. 2007;162:346–56. - PubMed
-
- Berardelli A, Rothwell JC, Hallett M, Thompson PD, Manfredi M, Marsden CD. The pathophysiology of primary dystonia. Brain. 1998;121:1195–212. - PubMed
-
- Breakefield XO, Blood AJ, Li Y, Hallett M, Hanson PI, Standaert DG. The pathophysiological basis of dystonias. Nat Rev Neurosci. 2008;9:222–34. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

