Inhibition of vascular calcium-gated chloride currents by blockers of KCa1.1, but not by modulators of KCa2.1 or KCa2.3 channels
- PMID: 19645713
- PMCID: PMC2757692
- DOI: 10.1111/j.1476-5381.2009.00332.x
Inhibition of vascular calcium-gated chloride currents by blockers of KCa1.1, but not by modulators of KCa2.1 or KCa2.3 channels
Abstract
Background and purpose: Recent pharmacological studies have proposed there is a high degree of similarity between calcium-activated Cl(-) channels (CaCCs) and large conductance, calcium-gated K(+) channels (K(Ca)1.1). The goal of the present study was to ascertain whether blockers of K(Ca)1.1 inhibited calcium-activated Cl(-) currents (I(ClCa)) and if the pharmacological overlap between K(Ca)1.1 and CaCCs extends to intermediate and small conductance, calcium-activated K(+) channels.
Experimental approaches: Whole-cell Cl(-) and K(+) currents were recorded from murine portal vein myocytes using the whole-cell variant of the patch clamp technique. CaCC currents were evoked by pipette solutions containing 500 nM free [Ca(2+)].
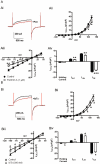
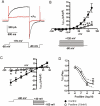
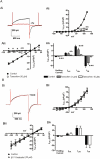
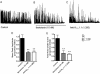
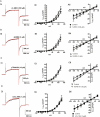
Key results: The selective K(Ca)1.1 blocker paxilline (1 microM) inhibited I(ClCa) by approximately 90%, whereas penitrem A (1 microM) and iberiotoxin (100 and 300 nM) reduced the amplitude of I(ClCa) by approximately 20%, as well as slowing channel deactivation. Paxilline also abolished the stimulatory effect of niflumic acid on the CaCC. In contrast, an antibody against the Ca(2+)-binding domain of murine K(Ca)1.1 had no effect on I(ClCa) while inhibiting spontaneous K(Ca)1.1 currents. Structurally different modulators of small and intermediate conductance calcium-activated K(+) channels (K(Ca)2.1 and K(Ca)2.3), namely 1-EBIO, (100 microM); NS309, (1 microM); TRAM-34, (10 microM); UCL 1684, (1 microM) had no effect on I(ClCa).
Conclusions and implications: These data show that the selective K(Ca)1.1 blockers also reduce I(ClCa) considerably. However, the pharmacological overlap that exists between CaCCs and K(Ca)1.1 does not extend to the calcium-binding domain or to other calcium-gated K(+) channels.
Figures
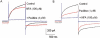
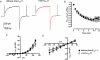
Similar articles
-
Cholesterol depletion alters amplitude and pharmacology of vascular calcium-activated chloride channels.Cardiovasc Res. 2010 Aug 1;87(3):476-84. doi: 10.1093/cvr/cvq057. Epub 2010 Feb 18. Cardiovasc Res. 2010. PMID: 20172862 Free PMC article.
-
Stimulation of Ca2+-gated Cl- currents by the calcium-dependent K+ channel modulators NS1619 [1,3-dihydro-1-[2-hydroxy-5-(trifluoromethyl)phenyl]-5-(trifluoromethyl)-2H-benzimidazol-2-one] and isopimaric acid.J Pharmacol Exp Ther. 2007 Jun;321(3):1075-84. doi: 10.1124/jpet.106.118786. Epub 2007 Mar 8. J Pharmacol Exp Ther. 2007. PMID: 17347326
-
Activation of chloride currents in murine portal vein smooth muscle cells by membrane depolarization involves intracellular calcium release.Am J Physiol Cell Physiol. 2005 Jan;288(1):C122-31. doi: 10.1152/ajpcell.00384.2004. Epub 2004 Sep 8. Am J Physiol Cell Physiol. 2005. PMID: 15355851
-
Pharmacology of Small- and Intermediate-Conductance Calcium-Activated Potassium Channels.Annu Rev Pharmacol Toxicol. 2020 Jan 6;60:219-240. doi: 10.1146/annurev-pharmtox-010919-023420. Epub 2019 Jul 23. Annu Rev Pharmacol Toxicol. 2020. PMID: 31337271 Free PMC article. Review.
-
Pharmacological gating modulation of small- and intermediate-conductance Ca(2+)-activated K(+) channels (KCa2.x and KCa3.1).Channels (Austin). 2015;9(6):336-43. doi: 10.1080/19336950.2015.1071748. Epub 2015 Jul 28. Channels (Austin). 2015. PMID: 26217968 Free PMC article. Review.
Cited by
-
Characterisation of K+ channels in human fetoplacental vascular smooth muscle cells.PLoS One. 2013;8(2):e57451. doi: 10.1371/journal.pone.0057451. Epub 2013 Feb 21. PLoS One. 2013. PMID: 23437391 Free PMC article.
-
Zinc pyrithione activates K+ channels and hyperpolarizes the membrane of rat pulmonary artery smooth muscle cells.PLoS One. 2018 Feb 23;13(2):e0192699. doi: 10.1371/journal.pone.0192699. eCollection 2018. PLoS One. 2018. PMID: 29474372 Free PMC article.
-
On the mode of action of emodepside: slow effects on membrane potential and voltage-activated currents in Ascaris suum.Br J Pharmacol. 2011 Sep;164(2b):453-70. doi: 10.1111/j.1476-5381.2011.01428.x. Br J Pharmacol. 2011. PMID: 21486286 Free PMC article.
-
Human spermatozoa possess a calcium-dependent chloride channel that may participate in the acrosomal reaction.J Physiol. 2012 Jun 1;590(11):2659-75. doi: 10.1113/jphysiol.2011.224485. Epub 2012 Apr 2. J Physiol. 2012. PMID: 22473777 Free PMC article.
-
Cholesterol depletion alters amplitude and pharmacology of vascular calcium-activated chloride channels.Cardiovasc Res. 2010 Aug 1;87(3):476-84. doi: 10.1093/cvr/cvq057. Epub 2010 Feb 18. Cardiovasc Res. 2010. PMID: 20172862 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

