Effects of fluticasone propionate dosage in an experimental model of feline asthma
- PMID: 19647461
- PMCID: PMC10911429
- DOI: 10.1016/j.jfms.2009.05.024
Effects of fluticasone propionate dosage in an experimental model of feline asthma
Abstract
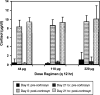
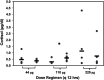
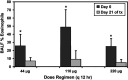
Cats with inflammatory bronchial disease are usually treated with glucocorticoid (GC) drugs to reduce airway inflammation. Inhalant GC delivery can preserve airway effects while systemic effects are minimized. An appropriate dosage regimen for inhaled GC in cats has not been investigated. A blinded, randomized, cross-over study design was used to investigate the ability of three different dosages of the inhalant GC fluticasone propionate delivered by metered dose inhaler to ameliorate eosinophilic airway inflammation in cats with experimentally induced allergic airway inflammation. Further, suppression of the hypothalamic-pituitary-adrenal axis (HPAA) at each dose was assessed. Fluticasone administered at dosages of 44, 110, or 220 microg q 12h reduced airway eosinophilia by 74%, 82%, or 81%, respectively (no difference). None of the dose regimens tested caused HPAA suppression. We conclude that a twice daily dosage of 44 microg fluticasone should be evaluated for the management of cats with naturally occurring inflammatory bronchial disease.
Copyright 2009 ESFM and AAFP. Published by Elsevier Ltd. All rights reserved.
Figures
Similar articles
-
Effects of high-dose inhaled fluticasone propionate on the hypothalamic-pituitary-adrenal axis in asthmatic patients with severely impaired lung function.Ann Allergy Asthma Immunol. 2004 Sep;93(3):253-8. doi: 10.1016/S1081-1206(10)61497-4. Ann Allergy Asthma Immunol. 2004. PMID: 15478385 Clinical Trial.
-
Diskus and diskhaler: efficacy and safety of fluticasone propionate via two dry powder inhalers in subjects with mild-to-moderate persistent asthma.Ann Allergy Asthma Immunol. 1999 Mar;82(3):273-80. doi: 10.1016/S1081-1206(10)62608-7. Ann Allergy Asthma Immunol. 1999. PMID: 10094218 Clinical Trial.
-
Effects of the inhaled corticosteroids fluticasone propionate, triamcinolone acetonide, and flunisolide and oral prednisone on the hypothalamic-pituitary-adrenal axis in adult patients with asthma.Clin Ther. 1999 Feb;21(2):353-67. doi: 10.1016/S0149-2918(00)88292-2. Clin Ther. 1999. PMID: 10211538 Clinical Trial.
-
Inhaled salmeterol/fluticasone propionate combination: a review of its use in persistent asthma.Drugs. 2000 Nov;60(5):1207-33. doi: 10.2165/00003495-200060050-00012. Drugs. 2000. PMID: 11129128 Review.
-
Inhaled fluticasone propionate: a review of its therapeutic efficacy at dosages < or = 500 microg/day in adults and adolescents with mild to moderate asthma.Drugs. 1999 May;57(5):769-803. doi: 10.2165/00003495-199957050-00016. Drugs. 1999. PMID: 10353302 Review.
Cited by
-
Role of animal models in biomedical research: a review.Lab Anim Res. 2022 Jul 1;38(1):18. doi: 10.1186/s42826-022-00128-1. Lab Anim Res. 2022. PMID: 35778730 Free PMC article. Review.
-
Breathe easy: inhalational therapy for feline inflammatory airway disease.J Feline Med Surg. 2023 Sep;25(9):1098612X231193054. doi: 10.1177/1098612X231193054. J Feline Med Surg. 2023. PMID: 37675792 Free PMC article. Review.
-
Treatment of naturally occurring asthma with inhaled fluticasone or oral prednisolone: A randomized pilot trial.Can J Vet Res. 2021 Jan;85(1):61-67. Can J Vet Res. 2021. PMID: 33390654 Free PMC article.
-
Noninvasive Recognition and Biomarkers of Early Allergic Asthma in Cats Using Multivariate Statistical Analysis of NMR Spectra of Exhaled Breath Condensate.PLoS One. 2016 Oct 20;11(10):e0164394. doi: 10.1371/journal.pone.0164394. eCollection 2016. PLoS One. 2016. PMID: 27764146 Free PMC article.
-
Comparison of pulmonary deposition of nebulized 99m technetium-diethylenetriamine-pentaacetic acid through 3 inhalation devices in healthy dogs.J Vet Intern Med. 2021 Mar;35(2):1080-1087. doi: 10.1111/jvim.16064. Epub 2021 Feb 24. J Vet Intern Med. 2021. PMID: 33624851 Free PMC article.
References
-
- Byers C.G., Dhupa N. Feline bronchial asthma: Treatment, Compend Contin Educ Pract Vet 27, 2005, 426–432.
-
- Padrid P. Feline asthma. Diagnosis and treatment, Vet Clin North Am Small Anim Pract 30, 2000, 1279–1293. - PubMed
-
- Cohn L.A. Glucocorticoids. Ettinger S.J., Feldman E.C. Textbook of veterinary internal medicine, 6th edn, 2005, WB Saunders: Philadelphia, 503–508.
-
- Allen D.B., Bielory L., Derendorf H., Dluhy R., Colice G.L., Szefler S.J. Inhaled corticosteroids: Past lessons and future issues, J Allergy Clin Immunol 112, 2003, S1–S40. - PubMed
-
- Dahl R. Systemic side effects of inhaled corticosteroids in patients with asthma, Respir Med 100, 2006, 1307–1317. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

