A loss-of-function polymorphism in the propeptide domain of the LOX gene and breast cancer
- PMID: 19654310
- PMCID: PMC2729564
- DOI: 10.1158/0008-5472.CAN-08-4818
A loss-of-function polymorphism in the propeptide domain of the LOX gene and breast cancer
Abstract
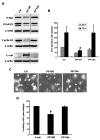
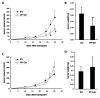
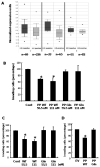
The lysyl oxidase (LOX) gene reverted Ras transformation of NIH 3T3 fibroblasts and tumor formation by gastric cancer cells, which frequently carry mutant RAS genes. The secreted lysyl oxidase proenzyme is processed to a propeptide (LOX-PP) and a functional enzyme (LOX). Unexpectedly, the tumor suppressor activity mapped to the LOX-PP domain, which inhibited tumor formation and the invasive phenotype of NF639 breast cancer cells driven by human epidermal growth factor receptor-2/neu, which signals via Ras. A single-nucleotide polymorphism, G473A (rs1800449), resulting in an Arg158Gln substitution in a highly conserved region within LOX-PP, occurs with an average 473A allele carrier frequency of 24.6% in the HapMap database, but was present in many breast cancer cell lines examined. Here, we show that the Arg-to-Gln substitution profoundly impairs the ability of LOX-PP to inhibit the invasive phenotype and tumor formation of NF639 cells in a xenograft model. LOX-PP Gln displayed attenuated ability to oppose the effects of LOX, which promoted a more invasive phenotype. In a case-control study of African American women, a potential association of the Gln-encoding A allele was seen with increased risk of estrogen receptor (ER)-alpha-negative invasive breast cancer in African American women. Consistently, LOX gene expression was higher in ER-negative versus ER-positive primary breast cancers, and LOX-PP Gln was unable to inhibit invasion by ER-negative cell lines. Thus, these findings identify for the first time genetic polymorphism as a mechanism of impaired tumor suppressor function of LOX-PP and suggest that it may play an etiologic role in ER-negative breast cancer.
Figures
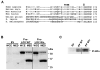

Similar articles
-
A polymorphism in the lysyl oxidase propeptide domain accelerates carcinogen-induced cancer.Carcinogenesis. 2018 Jul 3;39(7):921-930. doi: 10.1093/carcin/bgy045. Carcinogenesis. 2018. PMID: 29579155 Free PMC article.
-
The tumor suppressor activity of the lysyl oxidase propeptide reverses the invasive phenotype of Her-2/neu-driven breast cancer.Cancer Res. 2007 Feb 1;67(3):1105-12. doi: 10.1158/0008-5472.CAN-06-3867. Cancer Res. 2007. PMID: 17283144
-
Association of the G473A polymorphism and expression of lysyl oxidase with breast cancer risk and survival in European women: a hospital-based case-control study.PLoS One. 2014 Aug 20;9(8):e105579. doi: 10.1371/journal.pone.0105579. eCollection 2014. PLoS One. 2014. PMID: 25141126 Free PMC article.
-
Lysyl oxidases: a novel multifunctional amine oxidase family.Prog Nucleic Acid Res Mol Biol. 2001;70:1-32. doi: 10.1016/s0079-6603(01)70012-8. Prog Nucleic Acid Res Mol Biol. 2001. PMID: 11642359 Review.
-
Structural and functional diversity of lysyl oxidase and the LOX-like proteins.Biochim Biophys Acta. 2003 Apr 11;1647(1-2):220-4. doi: 10.1016/s1570-9639(03)00053-0. Biochim Biophys Acta. 2003. PMID: 12686136 Review.
Cited by
-
Activation of cellular chemotactic responses to chemokines coupled with oxidation of plasma membrane proteins by lysyl oxidase.J Neural Transm (Vienna). 2011 Jul;118(7):1091-9. doi: 10.1007/s00702-011-0642-5. Epub 2011 Apr 21. J Neural Transm (Vienna). 2011. PMID: 21509606
-
A function-blocking CD47 antibody suppresses stem cell and EGF signaling in triple-negative breast cancer.Oncotarget. 2016 Mar 1;7(9):10133-52. doi: 10.18632/oncotarget.7100. Oncotarget. 2016. PMID: 26840086 Free PMC article.
-
Oxidases and reactive oxygen species during hematopoiesis: a focus on megakaryocytes.J Cell Physiol. 2012 Oct;227(10):3355-62. doi: 10.1002/jcp.24071. J Cell Physiol. 2012. PMID: 22331622 Free PMC article. Review.
-
Inhibition of CIN85-mediated invasion by a novel SH3 domain binding motif in the lysyl oxidase propeptide.PLoS One. 2013 Oct 22;8(10):e77288. doi: 10.1371/journal.pone.0077288. eCollection 2013. PLoS One. 2013. PMID: 24167568 Free PMC article.
-
Silencing of hypoxia-inducible tumor suppressor lysyl oxidase gene by promoter methylation activates carbonic anhydrase IX in nasopharyngeal carcinoma.Am J Cancer Res. 2014 Nov 19;4(6):789-800. eCollection 2014. Am J Cancer Res. 2014. PMID: 25520868 Free PMC article.
References
-
- Kagan HM, Li W. Lysyl oxidase: properties, specificity, and biological roles inside and outside of the cell. J Cell Biochem. 2003;88:660–72. - PubMed
-
- Kagan HM, Trackman PC. Properties and function of lysyl oxidase. Am J Respir Cell Mol Biol. 1991;5:206–10. - PubMed
-
- Contente S, Kenyon K, Rimoldi D, Friedman RM. Expression of gene rrg is associated with reversion of NIH 3T3 transformed by LTR-c-H-ras. Science. 1990;249:796–8. - PubMed
-
- Kenyon K, Contente S, Trackman PC, Tang J, Kagan HM, Friedman RM. Lysyl oxidase and rrg messenger RNA. Science. 1991;253:802. - PubMed
-
- Bouez C, Reynaud C, Noblesse E, et al. The lysyl oxidase LOX is absent in basal and squamous cell carcinomas and its knockdown induces an invading phenotype in a skin equivalent model. Clin Cancer Res. 2006;12:1463–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

