Gonadotropin-releasing hormone (GnRH)-I and GnRH-II induce cell growth inhibition in human endometrial cancer cells: involvement of integrin beta3 and focal adhesion kinase
- PMID: 19656390
- PMCID: PMC2736964
- DOI: 10.1186/1477-7827-7-81
Gonadotropin-releasing hormone (GnRH)-I and GnRH-II induce cell growth inhibition in human endometrial cancer cells: involvement of integrin beta3 and focal adhesion kinase
Abstract
Endometrial carcinoma is the most common neoplasm of the female genital tract, accounting for nearly one half of all gynecologic cancers in the Western world. Although intensive research on pathological phenomena of endometrial cancer is currently going on, but exact cause and biological aspects of this disease are not well described yet. In addition to well-documented roles of gonadotropin-releasing hormone (GnRH) in hypopituitary ovarian (HPO) axis, the agonistic or antagonistic analogs (or both) of GnRH have been shown to inhibit the proliferation of a variety of human gynecologic cancers. Thus, in the present study, we further examined the possibility that GnRH induces integrin beta3 and activation of focal adhesion kinase (FAK) through mitogen-activated protein kinases (MAPKs), ERK1/2 and p38, to inhibit the growth of HEC1A endometrial cancer cell line. As a result, both GnRH-I and GnRH-II resulted in a significant increase in integrin beta3 expression and evoked the activation of FAK in a time-dependent manner in these cells. In addition, these analogs induced an activation of ERK1/2 and p38 MAPK in a time-dependent manner as downstream pathways of FAK. It appears that GnRH-II has much greater effect on the activation of FAK, ERK1/2 and p38 compared to GnRH-I in these cells. Further, we demonstrated that the growth inhibition of HEC1A cells by GnRH-I or GnRH-II is involved in the activation of integrin-FAK and ERK1/2 and p38 MAPK pathways. Taken together, these results suggest that GnRH may be involved in the inhibition of endometrial cancer cell growth via activation of integrin beta3 and FAK as a direct effect. This knowledge could contribute to a better understanding of the mechanisms implicated in the therapeutic action of GnRH and its biomedical application for the treatment against endometrial cancer.
Figures






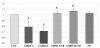
References
-
- Engel JB, Keller G, Schally AV, Nagy A, Chism DD, Halmos G. Effective treatment of experimental human endometrial cancers with targeted cytotoxic luteinizing hormone-releasing hormone analogues AN-152 and AN-207. Fertil Steril. 2005;83:1125–1133. doi: 10.1016/j.fertnstert.2004.10.042. - DOI - PubMed
-
- Savino L, Baldini B, Susini T, Pulli F, Antignani L, Massi GB. GnRH analogs in gynecological oncology: a review. J Chemother. 1992;4:312–320. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

