Spectrotemporal response properties of inferior colliculus neurons in alert monkey
- PMID: 19657026
- PMCID: PMC6666592
- DOI: 10.1523/JNEUROSCI.5459-08.2009
Spectrotemporal response properties of inferior colliculus neurons in alert monkey
Abstract
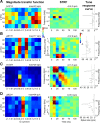
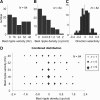
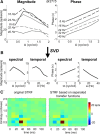
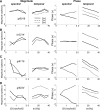
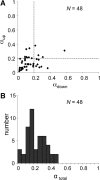
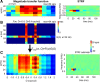
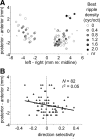
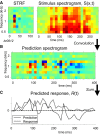
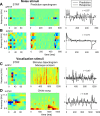
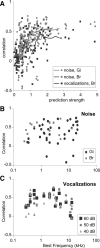
Because of its central position in the ascending auditory pathway, its large number of converging auditory brainstem inputs, and its fundamental role as a relay to auditory cortex and midbrain superior colliculus, the mammalian inferior colliculus (IC) is regarded pivotal for the integration of acoustic spectral-temporal cues to mediate sound-evoked behavior. However, detailed quantitative analyses of spectrotemporal neural responses are scarce. Moreover, most studies have been performed in anesthetized preparations, and it is unclear how to extrapolate findings to awake and behaving animals. Here, we characterize spectrotemporal receptive fields (STRFs) of single units in alert monkey IC by using a variety of broadband sounds with rippled amplitude spectra. We measured the response sensitivity to the ripple parameters density, Omega (cycles/octave), velocity, w (hertz), and direction selectivity, D. We observed a variety of dynamic STRFs, with a strong preference for low ripple densities, and a generally weak direction selectivity. Most cells preferred dynamic rippled stimuli above pure amplitude modulated noise (i.e., Omega = 0). Half of the cells could be characterized by good spectral-temporal separability, in which the ripple transfer function can be written as T(w, Omega) = F(w) x G(Omega). Inseparability could be attributed to a difference in responses to up and downward direction with respect to both amplitude and temporal phase. We tested linearity of IC neurons by using the STRF to predict neural responses to natural stimuli and broadband noise and discuss our results in the light of findings obtained from auditory cortex.
Figures





References
-
- Bour LJ, van Gisbergen JA, Bruijns J, Ottes FP. The double magnetic induction method for measuring eye movement: results in monkey and man. IEEE Trans Biomed Eng. 1984;31:419–427. - PubMed
-
- Chi T, Gao Y, Guyton MC, Ru P, Shamma S. Spectro-temporal modulation transfer functions and speech intelligibility. J Acoust Soc Am. 1999;106:2719–2732. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
