Human CD34/CD90 ASCs are capable of growing as sphere clusters, producing high levels of VEGF and forming capillaries
- PMID: 19657392
- PMCID: PMC2717331
- DOI: 10.1371/journal.pone.0006537
Human CD34/CD90 ASCs are capable of growing as sphere clusters, producing high levels of VEGF and forming capillaries
Abstract
Background: Human adult adipose tissue is an abundant source of mesenchymal stem cells (MSCs). Moreover, it is an easily accessible site producing a considerable amount of stem cells.
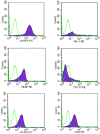
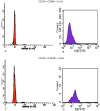
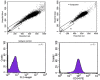
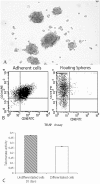
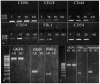
Methodology/principal findings: In this study, we have selected and characterized stem cells within the stromal vascular fraction (SVF) of human adult adipose tissue with the aim of understanding their differentiation capabilities and performance. We have found, within the SVF, different cell populations expressing MSC markers--including CD34, CD90, CD29, CD44, CD105, and CD117--and endothelial-progenitor-cell markers--including CD34, CD90, CD44, and CD54. Interestingly, CD34(+)/CD90(+) cells formed sphere clusters, when placed in non-adherent growth conditions. Moreover, they showed a high proliferative capability, a telomerase activity that was significantly higher than that found in differentiated cells, and contained a fraction of cells displaying the phenotype of a side population. When cultured in adipogenic medium, CD34(+)/CD90(+) quickly differentiated into adipocytes. In addition, they differentiated into endothelial cells (CD31(+)/VEGF(+)/Flk-1(+)) and, when placed in methylcellulose, were capable of forming capillary-like structures producing a high level of VEGF, as substantiated with ELISA tests.
Conclusions/significance: Our results demonstrate, for the first time, that CD34(+)/CD90(+) cells of human adipose tissue are capable of forming sphere clusters, when grown in free-floating conditions, and differentiate in endothelial cells that form capillary-like structures in methylcellulose. These cells might be suitable for tissue reconstruction in regenerative medicine, especially when patients need treatments for vascular disease.
Conflict of interest statement
Figures



References
-
- Verfaillie CM, Pera MF, Lansdorp PM. Stem cells: hype and reality. Hematology Am Soc Hematol Educ Program. 2002:369–391. - PubMed
-
- Conget PA, Minguell JJ. Phenotypical and functional properties of human bone marrow mesenchymal progenitor cells. J Cell Physiol. 1999;181:67–73. - PubMed
-
- Majumdar MK, Thiede MA, Mosca JD, Moorman M, Gerson SL. Phenotypic and functional comparison of cultures of marrow-derived mesenchymal stem cells (MSCs) and stromal cells. J Cell Physiol. 1998;176:57–66. - PubMed
-
- Schilling T, Noth U, Klein-Hitpass L, Jakob F, Schutze N. Plasticity in adipogenesis and osteogenesis of human mesenchymal stem cells. Mol Cell Endocrinol. 2007;271:1–17. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

