Population pharmacokinetics of sirolimus in de novo Chinese adult renal transplant patients
- PMID: 19660003
- PMCID: PMC2732940
- DOI: 10.1111/j.1365-2125.2009.03392.x
Population pharmacokinetics of sirolimus in de novo Chinese adult renal transplant patients
Abstract
Aims: This study was aimed at determining the population pharmacokinetics of sirolimus and identifying factors that explain pharmacokinetic variability in de novo Chinese adult renal transplant patients.
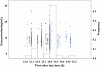
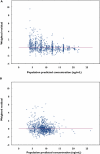
Methods: Data were retrospectively extracted from a formal multicentre clinical trial, which was originally designed to evaluate the safety and efficacy of cyclosporin dose reduction and cyclosporin elimination in patients receiving sirolimus. All patients received 12-month treatment, i.e. induction therapy with cyclosporin, sirolimus and corticosteroids during the first 3 months followed by either cyclosporin dose reduction or cyclosporin discontinuation thereafter. Eight-hundred and four sirolimus trough blood concentrations (C(0)) from 112 patients were used to develop a population pharmacokinetic model using the NONMEM program. A one-compartment model with first-order absorption and elimination was selected as the base model. The influence of demographic characteristics, biochemical and haematological indices, cyclosporin daily dose, cyclosporin C(0) as well as other commonly used co-medications were explored.
Results: The typical values with interindividual variability for apparent clearance (CL/F) and apparent volume of distribution (V/F) were 10.1 l h(-1) (23.8%) and 3670 l (56.7%), respectively. The residual variability was 29.9%. CL/F decreased significantly with silymarin or glycyrrhizin co-therapy in hepatically impaired patients, and with increasing total cholesterol levels or cyclosporin C(0). Moreover, CL/F increased nonlinearly with increasing sirolimus daily dose. The median parameter estimates from a nonparametric bootstrap procedure were comparable and within 5% of the estimates from NONMEM.
Conclusions: These results provide important information for clinicians to optimize sirolimus regimens in Chinese renal transplant patients.
Figures

Similar articles
-
Sirolimus population pharmacokinetic/pharmacogenetic analysis and bayesian modelling in kidney transplant recipients.Clin Pharmacokinet. 2006;45(11):1135-48. doi: 10.2165/00003088-200645110-00007. Clin Pharmacokinet. 2006. PMID: 17048977
-
Population pharmacokinetic characteristics of sirolimus in healthy Chinese subjects and renal transplant patients.Int J Clin Pharmacol Ther. 2016 Jun;54(6):433-41. doi: 10.5414/CP202499. Int J Clin Pharmacol Ther. 2016. PMID: 27117038 Clinical Trial.
-
Steady-state pharmacokinetics of sirolimus in stable adult Chinese renal transplant patients.Clin Pharmacol Drug Dev. 2014 May;3(3):235-41. doi: 10.1002/cpdd.96. Epub 2014 Feb 10. Clin Pharmacol Drug Dev. 2014. PMID: 27128614 Clinical Trial.
-
Safety and efficacy of TOR inhibitors in pediatric renal transplant recipients.Am J Kidney Dis. 2001 Oct;38(4 Suppl 2):S22-8. doi: 10.1053/ajkd.2001.27838. Am J Kidney Dis. 2001. PMID: 11583941 Review.
-
Clinical pharmacokinetics and therapeutic drug monitoring of sirolimus.Clin Ther. 2000;22 Suppl B:B101-121. doi: 10.1016/s0149-2918(00)89027-x. Clin Ther. 2000. PMID: 10823378 Review.
Cited by
-
The first study in pediatric: Population pharmacokinetics of sirolimus and its application in Chinese children with immune cytopenia.Int J Immunopathol Pharmacol. 2020 Jan-Dec;34:2058738420934936. doi: 10.1177/2058738420934936. Int J Immunopathol Pharmacol. 2020. PMID: 32720540 Free PMC article.
-
Exploring Sirolimus Pharmacokinetic Variability Using Data Available from the Routine Clinical Care of Renal Transplant Patients - Population Pharmacokinetic Approach.J Med Biochem. 2019 May 11;38(3):323-331. doi: 10.2478/jomb-2018-0030. eCollection 2019 Jul. J Med Biochem. 2019. PMID: 31156343 Free PMC article.
-
Remedial Dosing Recommendations for Sirolimus Delayed or Missed Dosages Caused by Poor Medication Compliance in Pediatric Tuberous Sclerosis Complex Patients.Curr Pharm Des. 2024;30(11):877-886. doi: 10.2174/0113816128299479240213151714. Curr Pharm Des. 2024. PMID: 38454763
-
Role of Statistical Random-Effects Linear Models in Personalized Medicine.Curr Pharmacogenomics Person Med. 2012 Mar;10(1):22-32. doi: 10.2174/1875692111201010022. Curr Pharmacogenomics Person Med. 2012. PMID: 23467392 Free PMC article.
-
Nonlinear population pharmacokinetics of sirolimus in patients with advanced cancer.CPT Pharmacometrics Syst Pharmacol. 2012 Dec 5;1(12):e17. doi: 10.1038/psp.2012.18. CPT Pharmacometrics Syst Pharmacol. 2012. PMID: 23887441 Free PMC article.
References
-
- Mahalati K, Kahan BD. Clinical pharmacokinetics of sirolimus. Clin Pharmacokinet. 2001;40:573–85. - PubMed
-
- Dumont FJ, Staruch MJ, Koprak SL, Melino MR, Sigal NH. Distinct mechanisms of suppression of murine T cell activation by the related macrolides FK-506 and rapamycin. J Immunol. 1990;144:251–8. - PubMed
-
- Kahan BD, Chang JY, Sehgal SN. Preclinical evaluation of a new potent immunosuppressive agent, rapamycin. Transplantation. 1991;52:185–91. - PubMed
-
- MacDonald AS. A worldwide, phase III, randomized, controlled, safety and efficacy study of a sirolimus/cyclosporine regimen for prevention of acute rejection in recipients of primary mismatched renal allografts. Transplantation. 2001;71:271–80. - PubMed
-
- Kreis H, Oberbauer R, Campistol JM, Mathew T, Daloze P, Schena FP, Burke JT, Brault Y, Gioud-Paquet M, Scarola JA, Neylan JF. Long-term benefits with sirolimus-based therapy after early cyclosporine withdrawal. J Am Soc Nephrol. 2004;15:809–17. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

