Regulation of monoamine oxidase A by the SRY gene on the Y chromosome
- PMID: 19661285
- PMCID: PMC2775015
- DOI: 10.1096/fj.09-139097
Regulation of monoamine oxidase A by the SRY gene on the Y chromosome
Abstract
Monoamine oxidase A (MAO A), encoded by the X chromosome, catalyzes the oxidative deamination of monoamine neurotransmitters, such as serotonin, and plays a critically important role in brain development and functions. Abnormal MAO A activity has been implicated in several neuropsychiatric disorders, such as depression, autism, and attention deficit hyperactivity disorder, which show sexual dimorphism. However, the molecular basis for these disease processes is unclear. Recently, we found that MAO A was a putative target gene directly regulated by a transcription factor encoded by the sex-determining region Y (SRY) gene located on the Y chromosome. We demonstrated that SRY activates both MAO A-promoter and catalytic activities in a human male neuroblastoma BE(2)C cell line. A functional SRY-binding site in the MAO A core promoter was identified and validated by electrophoretic mobility shift and chromatin immunoprecipitation (ChIP) analyses. Coimmunoprecipitation and ChIP assays showed that SRY and Sp1 form a transcriptional complex and synergistically activate MAO A transcription. This is the first study demonstrating that the Y-encoded transcription factor SRY is capable of regulating an X-located gene, suggesting a novel molecular mechanism for sexual dimorphism in neural development, brain functions, and initiation/progression of neural disorders associated with MAO A dysfunction.
Figures
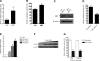


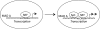
Similar articles
-
Transcription factor E2F-associated phosphoprotein (EAPP), RAM2/CDCA7L/JPO2 (R1), and simian virus 40 promoter factor 1 (Sp1) cooperatively regulate glucocorticoid activation of monoamine oxidase B.Mol Pharmacol. 2011 Feb;79(2):308-17. doi: 10.1124/mol.110.067439. Epub 2010 Oct 27. Mol Pharmacol. 2011. PMID: 20980443 Free PMC article.
-
Regulation of Monoamine Oxidase B Gene Expression: Key Roles for Transcription Factors Sp1, Egr1 and CREB, and microRNAs miR-300 and miR-1224.J Mol Biol. 2019 Mar 15;431(6):1127-1147. doi: 10.1016/j.jmb.2019.01.042. Epub 2019 Feb 7. J Mol Biol. 2019. PMID: 30738894
-
Dual functions of transcription factors, transforming growth factor-beta-inducible early gene (TIEG)2 and Sp3, are mediated by CACCC element and Sp1 sites of human monoamine oxidase (MAO) B gene.J Biol Chem. 2004 May 14;279(20):21021-8. doi: 10.1074/jbc.M312638200. Epub 2004 Mar 15. J Biol Chem. 2004. PMID: 15024015
-
Type A monoamine oxidase; its unique role in mood, behavior and neurodegeneration.J Neural Transm (Vienna). 2025 Mar;132(3):387-406. doi: 10.1007/s00702-024-02866-z. Epub 2024 Dec 2. J Neural Transm (Vienna). 2025. PMID: 39621110 Review.
-
Modulation of monoamine oxidase (MAO) expression in neuropsychiatric disorders: genetic and environmental factors involved in type A MAO expression.J Neural Transm (Vienna). 2016 Feb;123(2):91-106. doi: 10.1007/s00702-014-1362-4. Epub 2015 Jan 22. J Neural Transm (Vienna). 2016. PMID: 25604428 Review.
Cited by
-
The role of monoamine oxidase A in the neurobiology of aggressive, antisocial, and violent behavior: A tale of mice and men.Prog Neurobiol. 2020 Nov;194:101875. doi: 10.1016/j.pneurobio.2020.101875. Epub 2020 Jun 20. Prog Neurobiol. 2020. PMID: 32574581 Free PMC article. Review.
-
Amino acid function and docking site prediction through combining disease variants, structure alignments, sequence alignments, and molecular dynamics: a study of the HMG domain.BMC Bioinformatics. 2012 Mar 13;13 Suppl 2(Suppl 2):S3. doi: 10.1186/1471-2105-13-S2-S3. BMC Bioinformatics. 2012. PMID: 22536866 Free PMC article.
-
The human testis-determining factor SRY localizes in midbrain dopamine neurons and regulates multiple components of catecholamine synthesis and metabolism.J Neurochem. 2012 Jul;122(2):260-71. doi: 10.1111/j.1471-4159.2012.07782.x. Epub 2012 Jun 8. J Neurochem. 2012. PMID: 22568433 Free PMC article.
-
Expanding the toolbox of ADHD genetics. How can we make sense of parent of origin effects in ADHD and related behavioral phenotypes?Behav Brain Funct. 2015 Oct 16;11(1):33. doi: 10.1186/s12993-015-0078-4. Behav Brain Funct. 2015. PMID: 26475699 Free PMC article. Review.
-
Amphetamine manipulates monoamine oxidase-A level and behavior using theranostic aptamers of transcription factors AP-1/NF-kB.J Biomed Sci. 2016 Feb 3;23:21. doi: 10.1186/s12929-016-0239-2. J Biomed Sci. 2016. PMID: 26841904 Free PMC article.
References
-
- Lan N C, Heinzmann C, Gal A, Klisak I, Orth U, Lai E, Grimsby J, Sparkes R S, Mohandas T, Shih J C. Human monoamine oxidase A and B genes map to Xp 11.23 and are deleted in a patient with Norrie disease. Genomics. 1989;4:552–559. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

