Induction of protein body formation in plant leaves by elastin-like polypeptide fusions
- PMID: 19664215
- PMCID: PMC3224952
- DOI: 10.1186/1741-7007-7-48
Induction of protein body formation in plant leaves by elastin-like polypeptide fusions
Abstract
Background: Elastin-like polypeptides are synthetic biopolymers composed of a repeating pentapeptide 'VPGXG' sequence that are valuable for the simple non-chromatographic purification of recombinant proteins. In addition, elastin-like polypeptide fusions have been shown to enhance the accumulation of a range of different recombinant proteins in plants, thus addressing the major limitation of plant-based expression systems, which is a low production yield. This study's main objectives were to determine the general utility of elastin-like polypeptide protein fusions in various intracellular compartments and to elucidate elastin-like polypeptide's mechanism of action for increasing recombinant protein accumulation in the endoplasmic reticulum of plants.
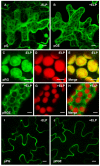
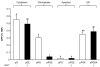
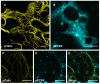
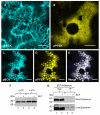
Results: The effect of elastin-like polypeptide fusions on the accumulation of green fluorescent protein targeted to the cytoplasm, chloroplasts, apoplast, and endoplasmic reticulum was evaluated. The endoplasmic reticulum was the only intracellular compartment in which an elastin-like polypeptide tag was shown to significantly enhance recombinant protein accumulation. Interestingly, endoplasmic reticulum-targeted elastin-like polypeptide fusions induced the formation of a novel type of protein body, which may be responsible for elastin-like polypeptide's positive effect on recombinant protein accumulation by excluding the heterologous protein from normal physiological turnover. Although expressed in the leaves of plants, these novel protein bodies appeared similar in size and morphology to the prolamin-based protein bodies naturally found in plant seeds. The elastin-like polypeptide-induced protein bodies were highly mobile organelles, exhibiting various dynamic patterns of movement throughout the cells, which were dependent on intact actin microfilaments and a functional actomyosin motility system.
Conclusion: An endoplasmic reticulum-targeted elastin-like polypeptide fusion approach provides an effective strategy for depositing large amounts of concentrated heterologous protein within the limited space of the cell via storage in stable protein bodies. Furthermore, encapsulation of recombinant proteins into physiologically inert organelles can function to insulate the protein from normal cellular mechanisms, thus limiting unnecessary stress to the host cell. Since elastin-like polypeptide is a mammalian-derived protein, this study demonstrates that plant seed-specific factors are not required for the formation of protein bodies in vegetative plant tissues, suggesting that the endoplasmic reticulum possesses an intrinsic ability to form protein body-like accretions in eukaryotic cells when overexpressing particular proteins.
Figures



Similar articles
-
Protein body formation in stable transgenic tobacco expressing elastin-like polypeptide and hydrophobin fusion proteins.BMC Biotechnol. 2013 May 10;13:40. doi: 10.1186/1472-6750-13-40. BMC Biotechnol. 2013. PMID: 23663656 Free PMC article.
-
Protein body-inducing fusions for high-level production and purification of recombinant proteins in plants.Plant Biotechnol J. 2011 May;9(4):419-33. doi: 10.1111/j.1467-7652.2011.00596.x. Epub 2011 Feb 22. Plant Biotechnol J. 2011. PMID: 21338467 Review.
-
Influence of elastin-like polypeptide and hydrophobin on recombinant hemagglutinin accumulations in transgenic tobacco plants.PLoS One. 2014 Jun 10;9(6):e99347. doi: 10.1371/journal.pone.0099347. eCollection 2014. PLoS One. 2014. PMID: 24914995 Free PMC article.
-
Production and characterization of in planta transiently produced polygalacturanase from Aspergillus niger and its fusions with hydrophobin or ELP tags.BMC Biotechnol. 2014 Jun 27;14:59. doi: 10.1186/1472-6750-14-59. BMC Biotechnol. 2014. PMID: 24970673 Free PMC article.
-
Green biofactories: recombinant protein production in plants.Recent Pat Biotechnol. 2010 Nov;4(3):242-59. Recent Pat Biotechnol. 2010. PMID: 21171961 Review.
Cited by
-
Tobacco, a highly efficient green bioreactor for production of therapeutic proteins.Biotechnol Adv. 2010 Mar-Apr;28(2):214-21. doi: 10.1016/j.biotechadv.2009.11.008. Epub 2009 Dec 2. Biotechnol Adv. 2010. PMID: 19961918 Free PMC article. Review.
-
A 5.2-kb insertion in the coding sequence of PavSCPL, a serine carboxypeptidase-like enhances fruit firmness in Prunus avium.Plant Biotechnol J. 2024 Jun;22(6):1622-1635. doi: 10.1111/pbi.14291. Epub 2024 Feb 28. Plant Biotechnol J. 2024. PMID: 38415985 Free PMC article.
-
Trafficking of endoplasmic reticulum-retained recombinant proteins is unpredictable in Arabidopsis thaliana.Front Plant Sci. 2014 Sep 15;5:473. doi: 10.3389/fpls.2014.00473. eCollection 2014. Front Plant Sci. 2014. PMID: 25309564 Free PMC article. Review.
-
N-Glycosylation of the SARS-CoV-2 Receptor Binding Domain Is Important for Functional Expression in Plants.Front Plant Sci. 2021 Jun 15;12:689104. doi: 10.3389/fpls.2021.689104. eCollection 2021. Front Plant Sci. 2021. PMID: 34211491 Free PMC article.
-
Plant-Produced Chimeric VHH-sIgA Against Enterohemorrhagic E. coli Intimin Shows Cross-Serotype Inhibition of Bacterial Adhesion to Epithelial Cells.Front Plant Sci. 2019 Mar 12;10:270. doi: 10.3389/fpls.2019.00270. eCollection 2019. Front Plant Sci. 2019. PMID: 30972081 Free PMC article.
References
-
- Stoger E, Ma JK, Fischer R, Christou P. Sowing the seeds of success: Pharmaceutical proteins from plants. Curr Opin Biotechnol. 2005;16:167–173. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

