Clinical features and outcome of patients with non-small-cell lung cancer who harbor EML4-ALK
- PMID: 19667264
- PMCID: PMC2744268
- DOI: 10.1200/JCO.2009.22.6993
Clinical features and outcome of patients with non-small-cell lung cancer who harbor EML4-ALK
Abstract
Purpose: The EML4-ALK fusion oncogene represents a novel molecular target in a small subset of non-small-cell lung cancers (NSCLC). To aid in identification and treatment of these patients, we examined the clinical characteristics and treatment outcomes of patients who had NSCLC with and without EML4-ALK.
Patients and methods: Patients with NSCLC were selected for genetic screening on the basis of two or more of the following characteristics: female sex, Asian ethnicity, never/light smoking history, and adenocarcinoma histology. EML4-ALK was identified by using fluorescent in situ hybridization for ALK rearrangements and was confirmed by immunohistochemistry for ALK expression. EGFR and KRAS mutations were determined by DNA sequencing.
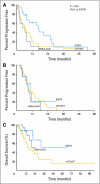
Results: Of 141 tumors screened, 19 (13%) were EML4-ALK mutant, 31 (22%) were EGFR mutant, and 91 (65%) were wild type (WT/WT) for both ALK and EGFR. Compared with the EGFR mutant and WT/WT cohorts, patients with EML4-ALK mutant tumors were significantly younger (P < .001 and P = .005) and were more likely to be men (P = .036 and P = .039). Patients with EML4-ALK-positive tumors, like patients who harbored EGFR mutations, also were more likely to be never/light smokers compared with patients in the WT/WT cohort (P < .001). Eighteen of the 19 EML4-ALK tumors were adenocarcinomas, predominantly the signet ring cell subtype. Among patients with metastatic disease, EML4-ALK positivity was associated with resistance to EGFR tyrosine kinase inhibitors (TKIs). Patients in the EML4-ALK cohort and the WT/WT cohort showed similar response rates to platinum-based combination chemotherapy and no difference in overall survival.
Conclusion: EML4-ALK defines a molecular subset of NSCLC with distinct clinical characteristics. Patients who harbor this mutation do not benefit from EGFR TKIs and should be directed to trials of ALK-targeted agents.
Conflict of interest statement
Authors' disclosures of potential conflicts of interest and author contributions are found at the end of this article.
Figures


Comment in
-
EML4-ALK: honing in on a new target in non-small-cell lung cancer.J Clin Oncol. 2009 Sep 10;27(26):4232-5. doi: 10.1200/JCO.2009.23.6661. Epub 2009 Aug 10. J Clin Oncol. 2009. PMID: 19667260 Free PMC article. No abstract available.
References
-
- Schiller JH, Harrington D, Belani CP, et al. Comparison of four chemotherapy regimens for advanced non–small-cell lung cancer. N Engl J Med. 2002;346:92–98. - PubMed
-
- Sandler A, Gray R, Perry MC, et al. Paclitaxel-carboplatin alone or with bevacizumab for non–small-cell lung cancer. N Engl J Med. 2006;355:2542–2550. - PubMed
-
- Lynch TJ, Bell DW, Sordella R, et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non–small-cell lung cancer to gefitinib. N Engl J Med. 2004;350:2129–2139. - PubMed
-
- Paez JG, Janne PA, Lee JC, et al. EGFR mutations in lung cancer: Correlation with clinical response to gefitinib therapy. Science. 2004;304:1497–1500. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

