Functional outcomes after multiple treatments with ranibizumab in neovascular age-related macular degeneration beyond visual acuity
- PMID: 19668506
- PMCID: PMC2704515
Functional outcomes after multiple treatments with ranibizumab in neovascular age-related macular degeneration beyond visual acuity
Abstract
Purpose: To evaluate neuroretinal function and anatomical outcomes in patients with neovascular age-related macular degeneration (AMD) after three treatments with ranibizumab.
Design: Observational case reports.
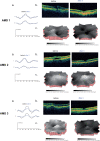
Methods: We investigated visual function in three patients, one female (80 years) and two male (77 and 74 years) with neovascular AMD. Twenty healthy participants served as control group. We measured visual acuity (Bailey-Lovie charts), contrast sensitivity (Pelli-Robson) and neuroretinal function using the multifocal electroretinogram (mfERG). Central macular thickness was evaluated using optical coherence tomography (OCT). Main outcome measures were central and peripheral mfERG peak to trough (N1P1) response density amplitudes and peak (P1) implicit times. All tests were performed before the first treatment (baseline) and after each of the three treatments with intravitreal 0.3 mg ranibizumab.
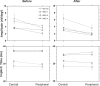
Results: Visual acuity and contrast sensitivity remained stable or improved. Central macular thickness decreased after three treatments in all three patients. We found no significant change in central and peripheral neuroretinal function in the AMD patients between pre- and post-treatments 2 and 3. Although the mfERG amplitudes in the AMD patients were not significantly reduced compared with the age-similar group at baseline, there was a statistically significant reduction in central and peripheral mfERG amplitudes after three treatments
Conclusion: Anatomical outcomes and central visual function improved or remained stable in the three AMD patients in concordance with past reports. Further investigations of possible adverse effects of ranibizumab on the central and peripheral neuroretina in large prospective clinical trials are suggested.
Keywords: OCT; age-related macular degeneration; multifocal ERG; multifocal electroretinogram; optical coherence tomography; ranibizumab.
Figures
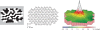

References
-
- Avery R, Pieramici D, Rabena M, Castellarin A, Nasir M, Guist M. Intravitreal bevacizumab (Avastin) for neovascular age-related macular degeneration. Ophthalmology. 2006;113:363–72. - PubMed
-
- Bailey I, Lovie J. New design principles for visual acuity letter charts. Am J Optom Physiol Opt. 1976;53:740–5. - PubMed
-
- Brown D, Kaiser P, Michels M, Soubrane G, Heier J, Kim R, Sy J, Schneider S, for the ANCHOR Study Group Ranibizumab versus verteporfin for neovascular Age-related macular degeneration. N Engl J Med. 2006;355:1432–44. - PubMed
-
- Costa R, Jorge R, Calucci D, Cardillo J, Melo LAJ, Scott I. Intravitreal bevacizumab for choroidal neovascularization caused by AMD (IBeNA Study): results of a phase 1 dose-escalation study. Invest Ophthalmol Vis Sci. 2006;47:4569–78. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

