The state of cholesterol metabolism in the liver of patients with primary biliary cirrhosis: the role of MDR3 expression
- PMID: 19669248
- PMCID: PMC2748377
- DOI: 10.1007/s12072-009-9137-y
The state of cholesterol metabolism in the liver of patients with primary biliary cirrhosis: the role of MDR3 expression
Abstract
Aim: Because dyslipidemia, such as hypercholesterolemia, is a characteristic of primary biliary cirrhosis (PBC), hepatic lipid metabolism may be disturbed in PBC patients. We examined the expression of lipid metabolism-associated genes in PBC liver.
Methods: All of the patients examined were in stage I or II PBC and without medication. RNA was isolated from liver specimens by needle biopsies of PBC patients and controls. The expression levels of various genes were measured by real-time RT-PCR. Multidrug resistance 3 (MDR3) expression was examined immunohistochemically. Statistical correlations between the gene expression levels and indices of blood testing were calculated.
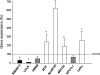
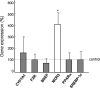
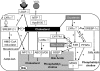
Results: The expression levels of sterol regulatory element-binding protein (SREBP) 2 and LDL receptor were significantly lower, and those of apolipoprotein B, microsomal triglyceride transfer protein, ATP-binding cassette G5, and liver X receptor α (LXRα) were significantly higher in the PBC liver than in the normal control liver. The expression levels of bile acid synthesis- and excretion-associated genes did not change, and those of farnesoid X receptor, peroxisome proliferator-activated receptor α, and SREBP-1c were similar between the PBC and normal liver. MDR3 gene expression levels in the PBC liver were more than 4-fold higher than those in the control liver. Immunohistochemically, strong canalicular staining for MDR3 was observed in the PBC liver. LXRα expression was positively correlated with MDR3 levels. Serum levels of γ-glutamyl transpeptidase (GGT) and IgM were negatively correlated with MDR3 levels.
Conclusions: Hepatocellular cholesterol metabolism was at least partially disturbed, even in the early stage of PBC. The most characteristic finding was a distinct elevation of MDR3 expression, and the MDR3 levels were negatively correlated with GGT and IgM levels.
Figures



References
-
- Wójcicka G, Jamroz-Wiśniewska A, Horoszewicz K, Bełtowski J. Liver X receptors (LXRs); part I: structure, function, regulation of activity, and role in lipid metabolism. Postepy Hig Med Dosw. 2007;61:736–759. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

