Entecavir for the treatment of lamivudine-refractory chronic hepatitis B patients in China
- PMID: 19669332
- PMCID: PMC2716831
- DOI: 10.1007/s12072-007-9016-3
Entecavir for the treatment of lamivudine-refractory chronic hepatitis B patients in China
Abstract
Purpose: This randomized, double-blind, placebo-controlled study was undertaken to evaluate the efficacy and safety of entecavir in Chinese patients with lamivudine-refractory chronic hepatitis B.
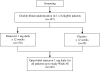
Methods: One hundred forty-five lamivudine-refractory patients with chronic hepatitis B were randomized to double-blind treatment with oral entecavir 1 mg (n = 116) or placebo (n = 29) daily for 12 weeks, followed by 36 weeks of open-label entecavir treatment. The primary efficacy endpoint was the mean change from baseline in serum hepatitis B virus (HBV) DNA by polymerase chain reaction (PCR) assay at week 12.
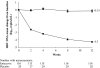
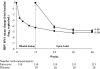
Results: At week 12, the mean change from baseline in serum HBV DNA by PCR assay was -4.30 log(10) copies/ml for patients on entecavir compared to -0.15 log(10 )copies/ml for patients on placebo (P < .0001). Among patients with baseline serum alanine aminotransferase (ALT) >1 x upper limit of normal (ULN), a higher proportion of entecavir than placebo patients (68% vs. 6%, respectively) achieved ALT normalization by week 12 (P < .0001). After 48 weeks of entecavir treatment, the mean change in HBV DNA by PCR assay was -5.08 log(10) copies/ml, and 85% of patients with baseline ALT >1 x ULN had achieved ALT normalization. The safety profile of entecavir was similar to that of placebo during the first 12 weeks of blinded dosing. Entecavir was also well tolerated during 36 weeks of open-label treatment.
Conclusions: Lamivudine-refractory chronic hepatitis B patients treated with entecavir demonstrated marked HBV DNA reduction and normalization of ALT in most cases. Entecavir treatment for 48 weeks was well tolerated.
Figures

Similar articles
-
Efficacy and safety of entecavir compared to lamivudine in nucleoside-naïve patients with chronic hepatitis B: a randomized double-blind trial in China.Hepatol Int. 2007 Sep;1(3):365-72. doi: 10.1007/s12072-007-9009-2. Epub 2007 Jul 31. Hepatol Int. 2007. PMID: 19669331 Free PMC article.
-
Efficacy and safety of entecavir in lamivudine-refractory patients with chronic hepatitis B: randomized controlled trial in Japanese patients.J Gastroenterol Hepatol. 2008 Sep;23(9):1320-6. doi: 10.1111/j.1440-1746.2008.05455.x. Epub 2008 Jun 28. J Gastroenterol Hepatol. 2008. PMID: 18554238 Clinical Trial.
-
A 24-week, parallel-group, open-label, randomized clinical trial comparing the early antiviral efficacy of telbivudine and entecavir in the treatment of hepatitis B e antigen-positive chronic hepatitis B virus infection in adult Chinese patients.Clin Ther. 2010 Apr;32(4):649-58. doi: 10.1016/j.clinthera.2010.04.001. Clin Ther. 2010. PMID: 20435234 Clinical Trial.
-
Cross-study analysis of the relative efficacies of oral antiviral therapies for chronic hepatitis B infection in nucleoside-naive patients.Clin Drug Investig. 2007;27(1):35-49. doi: 10.2165/00044011-200727010-00003. Clin Drug Investig. 2007. PMID: 17177578 Review.
-
Treatment of chronic hepatitis B: case selection and duration of therapy.J Gastroenterol Hepatol. 2002 Apr;17(4):409-14. doi: 10.1046/j.1440-1746.2002.02767.x. J Gastroenterol Hepatol. 2002. PMID: 11982721 Review.
Cited by
-
Safety and efficacy of entecavir for the treatment of chronic hepatitis B.Infect Drug Resist. 2011;4:55-64. doi: 10.2147/IDR.S4188. Epub 2011 Feb 3. Infect Drug Resist. 2011. PMID: 21694909 Free PMC article.
-
A comparison of treatment with adefovir and entecavir for chronic hepatitis B in China: The 2-year results of a prospective study: Adefovir versus Entercavir for Chronic Hepatitis B.Hepat Mon. 2011 Jan;11(1):27-31. Hepat Mon. 2011. PMID: 22087113 Free PMC article.
-
Entecavir: a review of its use in chronic hepatitis B.Drugs. 2009 May 29;69(8):1003-33. doi: 10.2165/00003495-200969080-00005. Drugs. 2009. PMID: 19496629 Review.
-
Clinical utility of entecavir for chronic hepatitis B in Chinese patients.Drug Des Devel Ther. 2013 Dec 12;8:13-24. doi: 10.2147/DDDT.S41423. Drug Des Devel Ther. 2013. PMID: 24376343 Free PMC article. Review.
-
The Discovery and Development of a Potent Antiviral Drug, Entecavir, for the Treatment of Chronic Hepatitis B.J Clin Transl Hepatol. 2013 Sep;1(1):51-8. doi: 10.14218/JCTH.2013.00006. Epub 2013 Sep 15. J Clin Transl Hepatol. 2013. PMID: 26357607 Free PMC article. Review.
References
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1111/j.1440-1746.2004.03728.x', 'is_inner': False, 'url': 'https://doi.org/10.1111/j.1440-1746.2004.03728.x'}]}
- Khan M, Dong JJ, Acharya SK, Dhagwahdorj Y, Abbas Z, Jafri W, et al. Hepatology issues in Asia: perspectives from regional leaders. J Gastroenterol Hepatol 2004;19:S419–30
-
- Hepatitis B Foundation [accessed 2005 Jan 19]. Available at: http://www.hepb.org/02-0360.hepb
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1002/jmv.10094', 'is_inner': False, 'url': 'https://doi.org/10.1002/jmv.10094'}, {'type': 'PubMed', 'value': '12116043', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/12116043/'}]}
- Sun Z, Ming L, Zhu X, Lu J. Prevention and control of hepatitis B in China. J Med Virol 2002;67:447–50 - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1016/S1089-3261(05)70170-7', 'is_inner': False, 'url': 'https://doi.org/10.1016/s1089-3261(05)70170-7'}, {'type': 'PubMed', 'value': '11385968', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/11385968/'}]}
- Lau GKK. Hepatitis B infection in China. Clin Liver Dis 2001;5:361–79 - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1111/j.1478-3231.2005.01134.x', 'is_inner': False, 'url': 'https://doi.org/10.1111/j.1478-3231.2005.01134.x'}, {'type': 'PubMed', 'value': '15910483', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/15910483/'}]}
- Liaw YF, Leung N, Guan R, Lau GKK, Merican I, McCaughan G, et al. Asian-Pacific consensus statement on the management of chronic hepatitis B: a 2005 update. Liver Int 2005;25:472–89 - PubMed
LinkOut - more resources
Full Text Sources
