Receptor tyrosine phosphatases control tracheal tube geometries through negative regulation of Egfr signaling
- PMID: 19675131
- PMCID: PMC2730367
- DOI: 10.1242/dev.033597
Receptor tyrosine phosphatases control tracheal tube geometries through negative regulation of Egfr signaling
Abstract
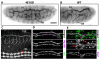
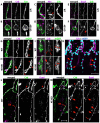
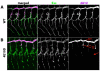
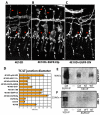
The formation of epithelial tubes with defined shapes and sizes is essential for organ development. We describe a unique tracheal tubulogenesis phenotype caused by loss of both Drosophila type III receptor tyrosine phosphatases (RPTPs), Ptp4E and Ptp10D. Ptp4E is the only widely expressed Drosophila RPTP, and is the last of the six fly RPTPs to be genetically characterized. We recently isolated mutations in Ptp4E, and discovered that, although Ptp4E null mutants have no detectable phenotypes, double mutants lacking both Ptp4E and Ptp10D display synthetic lethality at hatching owing to respiratory failure. In these double mutants, unicellular and terminal tracheal branches develop large bubble-like cysts that selectively incorporate apical cell surface markers. Cysts in unicellular branches are enlargements of the lumen that are sealed by adherens junctions, whereas cysts in terminal branches are cytoplasmic vacuoles. Cyst size and number are increased by tracheal expression of activated Egfr tyrosine kinase, and decreased by reducing Egfr levels. Ptp10D forms a complex with Egfr in transfected cells. Downregulation of Egfr signaling by the RPTPs is required for the construction of tubular lumens, whether extracellular or intracellular, by cells that undergo remodeling during branch morphogenesis. The Ptp4E Ptp10D phenotype represents the first evidence of an essential role for RPTPs in epithelial organ development. These findings might be relevant to organ development and disease in mammals, because PTPRJ (DEP-1), an ortholog of Ptp4E/Ptp10D, interacts with the hepatocyte growth factor receptor tyrosine kinase. PTPRJ corresponds to the murine Scc1 (suppressor of colon cancer) gene.
Figures



Similar articles
-
The novel Smad protein Expansion regulates the receptor tyrosine kinase pathway to control Drosophila tracheal tube size.Dev Biol. 2014 Sep 1;393(1):93-108. doi: 10.1016/j.ydbio.2014.06.016. Epub 2014 Jun 25. Dev Biol. 2014. PMID: 24973580 Free PMC article.
-
Redundancy and compensation in axon guidance: genetic analysis of the Drosophila Ptp10D/Ptp4E receptor tyrosine phosphatase subfamily.Neural Dev. 2008 Jan 31;3:3. doi: 10.1186/1749-8104-3-3. Neural Dev. 2008. PMID: 18237413 Free PMC article.
-
Interactions between Type III receptor tyrosine phosphatases and growth factor receptor tyrosine kinases regulate tracheal tube formation in Drosophila.Biol Open. 2012 Jun 15;1(6):548-58. doi: 10.1242/bio.2012471. Epub 2012 Apr 24. Biol Open. 2012. PMID: 23213447 Free PMC article.
-
R3 receptor tyrosine phosphatases: conserved regulators of receptor tyrosine kinase signaling and tubular organ development.Semin Cell Dev Biol. 2015 Jan;37:119-26. doi: 10.1016/j.semcdb.2014.09.005. Epub 2014 Sep 19. Semin Cell Dev Biol. 2015. PMID: 25242281 Free PMC article. Review.
-
Receptor protein-tyrosine phosphatase signalling in development.Int J Dev Biol. 1999;43(7):723-33. Int J Dev Biol. 1999. PMID: 10668981 Review.
Cited by
-
Interactions between a receptor tyrosine phosphatase and a cell surface ligand regulate axon guidance and glial-neuronal communication.Neuron. 2013 Jun 5;78(5):813-26. doi: 10.1016/j.neuron.2013.04.001. Neuron. 2013. PMID: 23764287 Free PMC article.
-
Drosophila Zpr1 (Zinc finger protein 1) is required downstream of both EGFR and FGFR signaling in tracheal subcellular lumen formation.PLoS One. 2012;7(9):e45649. doi: 10.1371/journal.pone.0045649. Epub 2012 Sep 18. PLoS One. 2012. PMID: 23029159 Free PMC article.
-
Epidermal Growth Factor Pathway Signaling in Drosophila Embryogenesis: Tools for Understanding Cancer.Cancers (Basel). 2017 Feb 7;9(2):16. doi: 10.3390/cancers9020016. Cancers (Basel). 2017. PMID: 28178204 Free PMC article. Review.
-
The ligand Sas and its receptor PTP10D drive tumour-suppressive cell competition.Nature. 2017 Feb 9;542(7640):246-250. doi: 10.1038/nature21033. Epub 2017 Jan 16. Nature. 2017. PMID: 28092921
-
An Ichor-dependent apical extracellular matrix regulates seamless tube shape and integrity.PLoS Genet. 2018 Jan 8;14(1):e1007146. doi: 10.1371/journal.pgen.1007146. eCollection 2018 Jan. PLoS Genet. 2018. PMID: 29309404 Free PMC article.
References
-
- Affolter, M. and Caussinus, E. (2008). Tracheal branching morphogenesis in Drosophila: new insights into cell behaviour and organ architecture. Development 135, 2055-2064. - PubMed
-
- Baumgartner, S., Littleton, J. T., Broadie, K., Bhat, M. A., Harbecke, R., Lengyel, J. A., Chiquet-Ehrismann, R., Prokop, A. and Bellen, H. J. (1996). A Drosophila neurexin is required for septate junction and blood-nerve barrier formation and function. Cell 87, 1059-1068. - PubMed
-
- Beitel, G. J. and Krasnow, M. A. (2000). Genetic control of epithelial tube size in the Drosophila tracheal system. Development 127, 3271-3282. - PubMed
-
- Buff, E., Carmena, A., Gisselbrecht, S., Jimenez, F. and Michelson, A. M. (1998). Signalling by the Drosophila epidermal growth factor receptor is required for the specification and diversification of embryonic muscle progenitors. Development 125, 2075-2086. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

