Enigmatic central canal contacting cells: immature neurons in "standby mode"?
- PMID: 19675235
- PMCID: PMC2753973
- DOI: 10.1523/JNEUROSCI.6183-08.2009
Enigmatic central canal contacting cells: immature neurons in "standby mode"?
Abstract
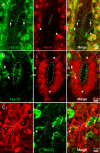
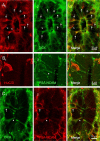
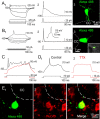
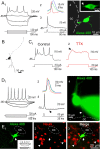
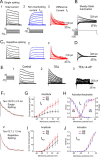
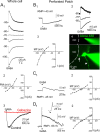
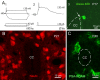
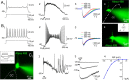
The region that surrounds the central canal of the spinal cord derives from the neural tube and retains a substantial degree of plasticity. In turtles, this region is a neurogenic niche where newborn neurons coexist with precursors, a fact that may be related with the endogenous repair capabilities of low vertebrates. Immunohistochemical evidence suggests that the ependyma of the mammalian spinal cord may contain cells with similar properties, but their actual nature remains unsolved. Here, we combined immunohistochemistry for cell-specific markers with patch-clamp recordings to test the hypothesis that the ependyma of neonatal rats contains immature neurons similar to those in low vertebrates. We found that a subclass of cells expressed HuC/D neuronal proteins, doublecortin, and PSA-NCAM (polysialylated neural cell adhesion molecule) but did not express NeuN (anti-neuronal nuclei). These immature neurons displayed electrophysiological properties ranging from slow Ca(2+)-mediated responses to fast repetitive Na(+) spikes, suggesting different stages of maturation. These cells originated in the embryo, because we found colocalization of neuronal markers with 5-bromo-2'-deoxyuridine when injected during embryonic day 7-17 but not in postnatal day 0-5. Our findings represent the first evidence that the ependyma of the rat spinal cord contains cells with molecular and functional features similar to immature neurons in adult neurogenic niches. The fact that these cells retain the expression of molecules that participate in migration and neuronal differentiation raises the possibility that the ependyma of the rat spinal cord is a reservoir of immature neurons in "standby mode," which under some circumstances (e.g., injury) may complete their maturation to integrate spinal circuits.
Figures



Similar articles
-
Spatial domains of progenitor-like cells and functional complexity of a stem cell niche in the neonatal rat spinal cord.Stem Cells. 2012 Sep;30(9):2020-31. doi: 10.1002/stem.1175. Stem Cells. 2012. PMID: 22821702 Free PMC article.
-
Postnatal maturation of mouse medullo-spinal cerebrospinal fluid-contacting neurons.Neuroscience. 2017 Feb 20;343:39-54. doi: 10.1016/j.neuroscience.2016.11.028. Epub 2016 Dec 7. Neuroscience. 2017. PMID: 27939302
-
A cell population that strongly expresses the CB1 cannabinoid receptor in the ependyma of the rat spinal cord.J Comp Neurol. 2013 Jan 1;521(1):233-51. doi: 10.1002/cne.23184. J Comp Neurol. 2013. PMID: 22791629
-
Brain Plasticity in Humans and Model Systems: Advances, Challenges, and Future Directions.Int J Mol Sci. 2021 Aug 28;22(17):9358. doi: 10.3390/ijms22179358. Int J Mol Sci. 2021. PMID: 34502267 Free PMC article. Review.
-
Progenitors in the Ependyma of the Spinal Cord: A Potential Resource for Self-Repair After Injury.Adv Exp Med Biol. 2017;1015:241-264. doi: 10.1007/978-3-319-62817-2_13. Adv Exp Med Biol. 2017. PMID: 29080030 Review.
Cited by
-
The Neural Stem Cell Properties of PKD2L1+ Cerebrospinal Fluid-Contacting Neurons in vitro.Front Cell Neurosci. 2021 Mar 15;15:630882. doi: 10.3389/fncel.2021.630882. eCollection 2021. Front Cell Neurosci. 2021. PMID: 33790741 Free PMC article.
-
A Role for the Cannabinoid 1 Receptor in Neuronal Differentiation of Adult Spinal Cord Progenitors in vitro is Revealed through Pharmacological Inhibition and Genetic Deletion.Front Neurosci. 2012 Jan 24;6:4. doi: 10.3389/fnins.2012.00004. eCollection 2012. Front Neurosci. 2012. PMID: 22291615 Free PMC article.
-
Roles of cerebrospinal fluid-contacting neurons as potential neural stem cells in the repair and regeneration of spinal cord injuries.Front Cell Dev Biol. 2024 Jun 25;12:1426395. doi: 10.3389/fcell.2024.1426395. eCollection 2024. Front Cell Dev Biol. 2024. PMID: 38983786 Free PMC article. Review.
-
Cholinergic Enhancement of Cell Proliferation in the Postnatal Neurogenic Niche of the Mammalian Spinal Cord.Stem Cells. 2015 Sep;33(9):2864-76. doi: 10.1002/stem.2077. Epub 2015 Jun 26. Stem Cells. 2015. PMID: 26038197 Free PMC article.
-
The dual developmental origin of spinal cerebrospinal fluid-contacting neurons gives rise to distinct functional subtypes.Sci Rep. 2017 Apr 7;7(1):719. doi: 10.1038/s41598-017-00350-1. Sci Rep. 2017. PMID: 28389647 Free PMC article.
References
-
- Alonso G. Neuronal progenitor-like cells expressing polysialylated neural cell adhesion molecule are present on the ventricular surface of the adult rat brain and spinal cord. J Comp Neurol. 1999;414:149–166. - PubMed
-
- Barry PH, Diamond JM. Junction potentials, electrode standard potentials, and other problems in interpreting electrical properties in membranes. J Membr Biol. 1970;3:93–122. - PubMed
-
- Beattie MS, Bresnahan JC, Komon J, Tovar CA, Van Meter M, Anderson DK, Faden AI, Hsu CY, Noble LJ, Salzman S, Young W. Endogenous repair after spinal cord contusion injuries in the rat. Exp Neurol. 1997;148:453–463. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous
