Recruitment in retractor bulbi muscle during eyeblink conditioning: EMG analysis and common-drive model
- PMID: 19675295
- PMCID: PMC2775390
- DOI: 10.1152/jn.00204.2009
Recruitment in retractor bulbi muscle during eyeblink conditioning: EMG analysis and common-drive model
Abstract
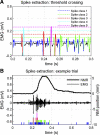
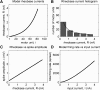
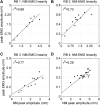
To analyze properly the role of the cerebellum in classical conditioning of the eyeblink and nictitating membrane (NM) response, the control of conditioned response dynamics must be better understood. Previous studies have suggested that the control signal is linearly related to the CR as a result of recruitment within the accessory abducens motoneuron pool, which acts to linearize retractor bulbi muscle and NM response mechanics. Here we investigate possible recruitment mechanisms. Data came from simultaneous recordings of NM position and multiunit electromyographic (EMG) activity from the retractor bulbi muscle of rabbits during eyeblink conditioning, in which tone and periocular shock act as conditional and unconditional stimuli, respectively. Action potentials (spikes) were extracted and classified by amplitude. Firing rates of spikes with different amplitudes were analyzed with respect to NM response temporal profiles and total EMG spike firing rate. Four main regularities were revealed and quantified: 1) spike amplitude increased with response amplitude; 2) smaller spikes always appeared before larger spikes; 3) subsequent firing rates covaried for spikes of different amplitude, with smaller spikes always firing at higher rates than larger ones; and 4) firing-rate profiles were approximately Gaussian for all amplitudes. These regularities suggest that recruitment does take place in the retractor bulbi muscle during conditioned NM responses and that all motoneurons receive the same command signal (common-drive hypothesis). To test this hypothesis, a model of the motoneuron pool was constructed in which motoneurons had a range of intrinsic thresholds distributed exponentially, with threshold linearly related to EMG spike amplitude. Each neuron received the same input signal as required by the common-drive assumption. This simple model reproduced the main features of the data, suggesting that conditioned NM responses are controlled by a common-drive mechanism that enables simple commands to determine response topography in a linear fashion.
Figures





Similar articles
-
Evidence from retractor bulbi EMG for linearized motor control of conditioned nictitating membrane responses.J Neurophysiol. 2007 Oct;98(4):2074-88. doi: 10.1152/jn.00210.2007. Epub 2007 Jul 5. J Neurophysiol. 2007. PMID: 17615135
-
Discharge profiles of abducens, accessory abducens, and orbicularis oculi motoneurons during reflex and conditioned blinks in alert cats.J Neurophysiol. 1999 Apr;81(4):1666-84. doi: 10.1152/jn.1999.81.4.1666. J Neurophysiol. 1999. PMID: 10200203
-
Localization of retractor bulbi motoneurons in the rabbit.Brain Res. 1981 Dec 7;226(1-2):93-106. doi: 10.1016/0006-8993(81)91085-4. Brain Res. 1981. PMID: 7296302
-
Physiology of the eyelid motor system.Ann N Y Acad Sci. 2003 Oct;1004:1-9. doi: 10.1196/annals.1303.001. Ann N Y Acad Sci. 2003. PMID: 14662442 Review.
-
The role of interpositus nucleus in eyelid conditioned responses.Cerebellum. 2002 Dec;1(4):289-308. doi: 10.1080/147342202320883597. Cerebellum. 2002. PMID: 12879967 Review.
Cited by
-
An agonist-antagonist cerebellar nuclear system controlling eyelid kinematics during motor learning.Front Neuroanat. 2012 Mar 14;6:8. doi: 10.3389/fnana.2012.00008. eCollection 2012. Front Neuroanat. 2012. PMID: 22435053 Free PMC article.
-
Adaptive timing of motor output in the mouse: the role of movement oscillations in eyelid conditioning.Front Integr Neurosci. 2011 Nov 30;5:72. doi: 10.3389/fnint.2011.00072. eCollection 2011. Front Integr Neurosci. 2011. PMID: 22144951 Free PMC article.
-
Cerebellar plasticity and the automation of first-order rules.J Neurosci. 2011 Feb 9;31(6):2305-12. doi: 10.1523/JNEUROSCI.4358-10.2011. J Neurosci. 2011. PMID: 21307266 Free PMC article.
-
Sensory prediction or motor control? Application of marr-albus type models of cerebellar function to classical conditioning.Front Comput Neurosci. 2010 Oct 4;4:140. doi: 10.3389/fncom.2010.00140. eCollection 2010. Front Comput Neurosci. 2010. PMID: 21031161 Free PMC article.
-
Bidirectional learning in upbound and downbound microzones of the cerebellum.Nat Rev Neurosci. 2021 Feb;22(2):92-110. doi: 10.1038/s41583-020-00392-x. Epub 2020 Nov 17. Nat Rev Neurosci. 2021. PMID: 33203932 Review.
References
-
- Aghajanian GK, McCall RB. Serotonergic synaptic input to facial motoneurons: localization by electron-microscopic autoradiography. Neuroscience 5: 2155–2162, 1980 - PubMed
-
- Akaboshi K, Masakado Y, Chino N. Quantitative EMG and motor unit recruitment threshold using a concentric needle with quadrifilar electrode. Muscle Nerve 23: 361–367, 2000 - PubMed
-
- Alvarado J, Steinacker A, Bach-y-Rita P. The ultrastructure of the retractor bulbi muscle of the cat (Abstract). Invest Ophthalmol 6: 548, 1967
-
- Angelaki DE, Hess BJM. Control of eye orientation: where does the brain's role end and the muscle's begin? Eur J Neurosci 19: 1–10, 2004 - PubMed
-
- Attwell PJ, Ivarsson M, Millar L, Yeo CH. Cerebellar mechanisms in eyeblink conditioning. Ann NY Acad Sci 978: 79–92, 2002 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

