Stress-induced potentiation of cocaine reward: a role for CRF R1 and CREB
- PMID: 19675537
- PMCID: PMC4034179
- DOI: 10.1038/npp.2009.91
Stress-induced potentiation of cocaine reward: a role for CRF R1 and CREB
Abstract
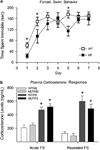
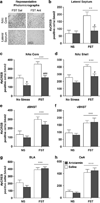
Both clinical and preclinical research have shown that stress can potentiate drug use; however, the underlying mechanisms of this interaction are unknown. Previously, we have shown that a single exposure to forced swim (FS) reinstates extinguished conditioned place preference (CPP) to cocaine and that cAMP response element binding protein (CREB) is necessary for this response. CREB can be activated by corticotropin releasing factor (CRF) receptor type 1 (CRF(R1)) binding, which mediates neuroendocrine and behavioral responses to stress as well as to drugs of abuse. The present experiments investigate whether changes in cocaine reward elicited by previous exposure to stress are mediated by CREB and/or CRF(R1). Chronic exposure to FS in advance of conditioning enhances cocaine CPP in wild-type mice, but this is blocked in CREB-deficient mice. In addition, pretreatment with the CRF(R1) antagonist, antalarmin, before FS exposure blocks this stress-induced enhancement of cocaine CPP. Furthermore, FS-induced increase in phosphorylated CREB (pCREB), specifically in the lateral septum (LS) and nucleus accumbens (NAc) is also blocked by antalarmin. Taken together, these studies suggest that both CREB and CRF(R1) activation are necessary for stress-induced potentiation of drug reward.
Conflict of interest statement
Figures




Similar articles
-
Beta-2 adrenergic receptors mediate stress-evoked reinstatement of cocaine-induced conditioned place preference and increases in CRF mRNA in the bed nucleus of the stria terminalis in mice.Psychopharmacology (Berl). 2014 Oct;231(20):3953-63. doi: 10.1007/s00213-014-3535-0. Epub 2014 Apr 3. Psychopharmacology (Berl). 2014. PMID: 24696080 Free PMC article.
-
Social interaction reward in rats has anti-stress effects.Addict Biol. 2021 Jan;26(1):e12878. doi: 10.1111/adb.12878. Epub 2020 Jan 26. Addict Biol. 2021. PMID: 31984611 Free PMC article.
-
cAMP response element-binding protein is required for stress but not cocaine-induced reinstatement.J Neurosci. 2004 Jul 28;24(30):6686-92. doi: 10.1523/JNEUROSCI.1706-04.2004. J Neurosci. 2004. PMID: 15282271 Free PMC article.
-
Reflections on: "A general role for adaptations in G-Proteins and the cyclic AMP system in mediating the chronic actions of morphine and cocaine on neuronal function".Brain Res. 2016 Aug 15;1645:71-4. doi: 10.1016/j.brainres.2015.12.039. Epub 2015 Dec 29. Brain Res. 2016. PMID: 26740398 Free PMC article. Review.
-
Roles of nucleus accumbens CREB and dynorphin in dysregulation of motivation.Cold Spring Harb Perspect Med. 2013 Feb 1;3(2):a012005. doi: 10.1101/cshperspect.a012005. Cold Spring Harb Perspect Med. 2013. PMID: 23293139 Free PMC article. Review.
Cited by
-
The behavioral, anatomical and pharmacological parallels between social attachment, love and addiction.Psychopharmacology (Berl). 2012 Nov;224(1):1-26. doi: 10.1007/s00213-012-2794-x. Epub 2012 Aug 11. Psychopharmacology (Berl). 2012. PMID: 22885871 Free PMC article. Review.
-
Repeated social defeat stress enhances glutamatergic synaptic plasticity in the VTA and cocaine place conditioning.Elife. 2016 Jul 4;5:e15448. doi: 10.7554/eLife.15448. Elife. 2016. PMID: 27374604 Free PMC article.
-
Corticotropin-Releasing Factor in the Nucleus Accumbens Does Not Drive High Levels of Cocaine Consumption.bioRxiv [Preprint]. 2025 Feb 26:2025.02.25.640140. doi: 10.1101/2025.02.25.640140. bioRxiv. 2025. PMID: 40060554 Free PMC article. Preprint.
-
Genetic Tagging Uncovers a Robust, Selective Activation of the Thalamic Paraventricular Nucleus by Adverse Experiences Early in Life.Biol Psychiatry Glob Open Sci. 2023 Jan 24;3(4):746-755. doi: 10.1016/j.bpsgos.2023.01.002. eCollection 2023 Oct. Biol Psychiatry Glob Open Sci. 2023. PMID: 37881549 Free PMC article.
-
Effects of acute stress on acquisition of nicotine conditioned place preference in adolescent rats: a role for corticotropin-releasing factor 1 receptors.Psychopharmacology (Berl). 2012 Jan;219(1):73-82. doi: 10.1007/s00213-011-2378-1. Epub 2011 Jul 1. Psychopharmacology (Berl). 2012. PMID: 21720754
References
-
- Carlezon WA, Jr, Thome J, Olson VG, Lane-Ladd SB, Brodkin ES, Hiroi N, Duman RS, Neve RL, Nestler EJ. Regulation of cocaine reward by CREB. Science. 1998;282:2272–2275. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

