Parabrachial coding of sapid sucrose: relevance to reward and obesity
- PMID: 19686159
- PMCID: PMC3607513
- DOI: 10.1111/j.1749-6632.2009.03930.x
Parabrachial coding of sapid sucrose: relevance to reward and obesity
Abstract
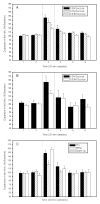
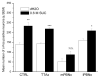
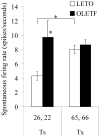
Cumulative evidence in rats suggests that the pontine parabrachial nuclei (PBN) are necessary for assigning hedonic value to taste stimuli. In a series of studies, our laboratory has investigated the parabrachial coding of sapid sucrose in normal and obese rats. First, using chronic microdialysis, we demonstrated that sucrose intake increases dopamine release in the nucleus accumbens, an effect that is dependent on oral stimulation and on concentration. The dopamine response was independent of the thalamocortical gustatory system but was blunted substantially by lesions of the PBN. Similar lesions of the PBN but not the thalamic taste relay diminished cFos activation in the nucleus accumbens caused by sucrose ingestion. Recent single-neuron recording studies have demonstrated that processing of sucrose-evoked activity in the PBN is altered in Otsuka Long Evans Tokushima Fatty (OLETF) rats, which develop obesity due to chronic overeating and express increased avidity to sweet. Compared with lean controls, taste neurons in OLETF rats had reduced overall sensitivity to sucrose and altered concentration responses, with decreased responses to lower concentrations and augmented responses to higher concentrations. The decreased sensitivity to sucrose was specific to NaCl-best neurons that also responded to sucrose, but the concentration effects were carried by the sucrose-specific neurons. Collectively, these findings support the hypothesis that the PBN enables taste stimuli to engage the reward system and, in doing so, influences food intake and body weight regulation. Obesity, in turn, may further alter the gustatory code via forebrain connections to the taste relays or hormonal changes consequent to weight gain.
Figures


References
-
- Norgren R, et al. Motivational modulation of taste. In: Ono T, M. G, Llinas R, Berthoz A, Norgren R, Nishijo H, Tamura R, editors. Cognition and Emotion in the Brain. Vol. International Congress Series. Elsevier; New York: 2003. pp. 319–334.
-
- Lundy RFJ, Norgren R. Gustatory System. In: Paxinos G, editor. The Rat Nervous System. Elsevier Academic Press; San Diego, CA: 2004. pp. 891–921.
-
- Smith GP. Accumbens dopamine mediates the rewarding effect of orosensory stimulation by sucrose. Appetite. 2004;43:11–3. - PubMed
-
- Wang GJ, Volkow ND, Fowler JS. The role of dopamine in motivation for food in humans: implications for obesity. Expert Opin Ther Targets. 2002;6:601–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

