Transgene expression is associated with copy number and cytomegalovirus promoter methylation in transgenic pigs
- PMID: 19688097
- PMCID: PMC2723931
- DOI: 10.1371/journal.pone.0006679
Transgene expression is associated with copy number and cytomegalovirus promoter methylation in transgenic pigs
Abstract
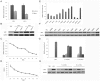
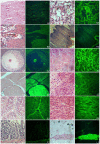
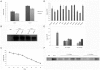
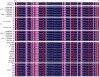
Transgenic animals have been used for years to study gene function, produce important proteins, and generate models for the study of human diseases. However, inheritance and expression instability of the transgene in transgenic animals is a major limitation. Copy number and promoter methylation are known to regulate gene expression, but no report has systematically examined their effect on transgene expression. In the study, we generated two transgenic pigs by somatic cell nuclear transfer (SCNT) that express green fluorescent protein (GFP) driven by cytomegalovirus (CMV). Absolute quantitative real-time PCR and bisulfite sequencing were performed to determine transgene copy number and promoter methylation level. The correlation of transgene expression with copy number and promoter methylation was analyzed in individual development, fibroblast cells, various tissues, and offspring of the transgenic pigs. Our results demonstrate that transgene expression is associated with copy number and CMV promoter methylation in transgenic pigs.
Conflict of interest statement
Figures
References
-
- Houdebine LM. The methods to generate transgenic animals and to control transgene expression. Biotechnology. 2002;98:145–160. - PubMed
-
- Park KW, Cheong HT, Lai L, Im GS, Kühholzer B, et al. Production of nuclear transfer-derived swine that express the enhanced green fluorescent protein. Anim Biotechnol. 2001;12:173–181. - PubMed
-
- Phelps CJ, Koike C, Vaught TD, Kühholzer B, Samuel M, et al. A transgenic pig expressing the enhanced green fluorescent protein produced by nuclear transfer using colchicine-treated fibroblasts as donor cells. Science. 2003;299:411–414.
-
- Gubin AN, Reddy B, Njoroge JM, Miller JL. Long-term Retroviral-mediated stable expression of Green Fluorescent Protein in mammalian cells. Biochem Biophys Res Commun. 1997;236:347–350. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

